Translate this page into:
High-Sensitivity C-Reactive Protein-to-Albumin Ratio in Predicting the Major Adverse Cardiovascular Event in Acute Coronary Syndrome at Presentation
*Corresponding author: Sajad Rashid Mir, Department of Cardiology, Nizam’s Institute of Medical Sciences, Hyderabad, Telangana, India. mir.sajadrashid007@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Mir SR, Lakshmi VS. High-sensitivity C-reactive protein-to-albumin ratio in predicting the major adverse cardiovascular event in acute coronary syndrome at presentation. Indian J Cardiovasc Dis Women 2022;7:130-6.
Abstract
Objective:
Our study aimed to determine the association between high sensitivity-c reactive protein to albumin ratio (CAR) and in-hospital major adverse cardiovascular event (MACE) in patients with acute coronary syndrome and also to assess whether hs-CAR is a better marker than hsCRP or albumin alone in predicting MACE.
Materials and Methods:
We enrolled 110 cases who were hospitalized and major adverse cardiovascular event was defined as cardiogenic shock, acute heart failure, reinfarction and death. Blood sample for Serum albumin and hs-crp was taken at the time when patients were admitted.
Results:
The incidence of MACE was more in patients with high CAR (≥1.8 group) as compared to those with low CAR (<1.8 group).
Conclusion:
The CAR is an independent predictor of MACE in patients who present with ACS.
Keywords
Acute coronary syndrome
High-sensitivity C reactive protein
Serum albumin
High-sensitivity C-reactive protein-to-albumin ratio
Major adverse cardiovascular event
ABSTRACT IMAGE
INTRODUCTION
It is a well-known fact that the atherosclerosis sets in with the onset of inflammation.[1] Inflammation has been proposed to be an essential determinant for adverse event in coronary artery disease patients. There are many biomarkers which have been studied for the diagnosis and risk stratification of patients with acute coronary syndrome (ACS). The levels of acute-phase reactant proteins, such as hs-CRP and albumin, have an important bearing on the severity of inflammatory response.[2,3]
C-reactive protein (CRP) is an acute phase reactant and belongs to the pentraxin family of proteins. It is produced by the liver whenever there is an inflammatory response. Acute infections, inflammatory states, and trauma lead to increase in the levels of this protein. Some newer techniques such as immunoturbidimetry, immunonephelometry, high-sensitivity enzyme-linked immunosorbent assay, and resonant acoustic profiling are used to estimate CRP with very high sensitivity (0.01–10 mg/l).[4] Low grades of systemic inflammation can be quantified with the help these high-sensitivity assays, even in the absence of overt systemic inflammatory disorders.
Both hs-CRP and albumin are produced by hepatocytes, and there is an inverse relationship of albumin with ACS. Thus, the hs-CRP-to-albumin ratio (CAR) that is suggestive of the balance of hs-CRP and albumin may give more sensitive results in assessing the inflammatory status than the use of either marker alone. There have been many studies that revealed that hs-CAR can be used as a novel inflammatory prognostic marker in some malignancies.[5-8] Many studies are there which have proposed a relation between hs-CAR and cardiovascular diseases.[9-11] Moreover, there is also an association between hs-CAR and severity of CAD in patients with ACS[10] and stable angina.[11]
However, till date, there have been fewer studies to reveal the association between hs-CAR and major adverse cardiac events (MACEs) such as acute heart failure, cardiogenic shock, reinfarction, and death in ACS. Therefore, the main objective of our study was to assess whether hs-CAR could be used to predict in-hospital MACEs in patients having ACS.
MATERIAL AND METHODS
Study population
It was a prospective observational study and patients with ACS who were admitted consecutively to the Department of Cardiology, NIMS, from October to December 2021, were enrolled in our study. ACS was diagnosed according to ACCF /AHA guidelines[12,13] The patients who had age >18 years, with diagnosis of ACS, and showed willingness to participate in the study were included in the study.
Patients who had eGFR <15 ml/kg /m2, liver diseases, malignancy, chronic heart failure (NYHA class >II), valvular heart disease, current acute stroke, venous thromboembolism, and prior history of thrombolysis were excluded from the study. This study was approved by the Ethics Committee of Nizam’s Institute of medical sciences and complied with the Declaration of Helsinki. Total of 110 among the patients included in the study had complete data and were enrolled for analysis.
Blood samples were collected at the time of admission even before giving heparin or thrombolysis treatment. Detailed history was taken and the risk factors, including hypertension, diabetes mellitus, obesity, OSA, and smoking status, were documented. Detailed clinical examination was also done. Blood biochemical analysis was carried out. It included lipid profile, blood sugar, hs-CRP and albumin levels, and liver and renal function. The biochemical analyzer (Beckman Coulter AU5800, CA, USA) was used to measure the hs-CRP and albumin levels the clinical laboratory as per the manufacturer’s instructions.
In-hospital MACE was regarded as the end point of our study. It was defined as acute heart failure, cardiogenic shock, reinfarction, and all-cause death as per the clinical studies by Hartopo et al.[14]
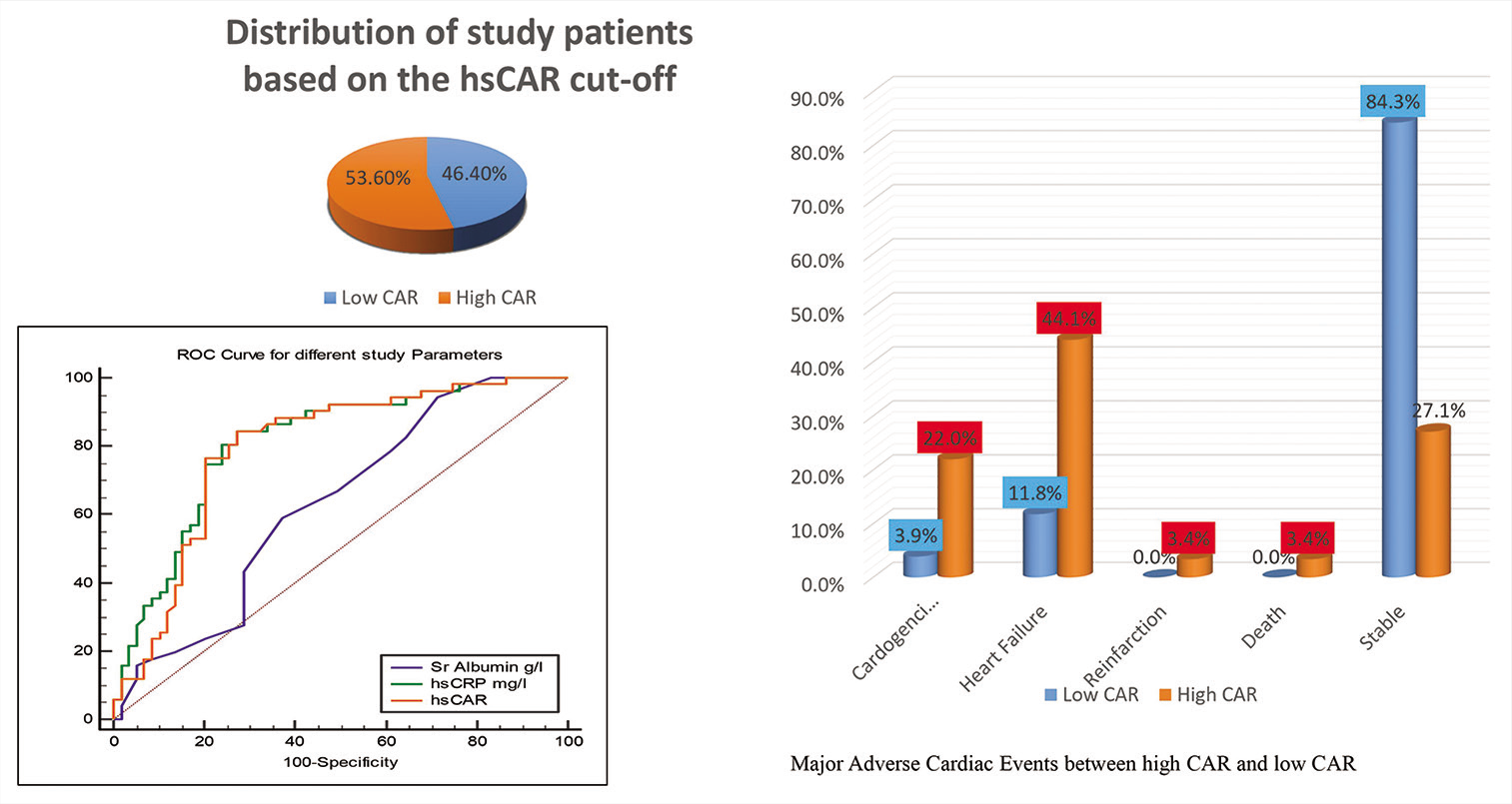
To perform statistical analyses, Statistical Package for the Social Sciences for Windows, Version 22.0. released in 2013. Armonk, NY: IBM Corp was used. Study patients having CAR <1.8 were labeled as “low” CAR group and CAR >1.8 as “high” CAR group as per the receiver operating characteristic (ROC) curve. The comparison was drawn between the two groups based on the baseline characteristics and MACE incidence rates.
ROC curve analysis was performed for hs-CAR for demonstrating the cutoff between low and high CAR patients. For comparing the mean age (in years) based on CAR and MACE, Mann—Whitney Test was used. To compare the gender and risk factors based on CAR and MACE, Chi-Square test was used. Logistic regression analysis model was performed for predicting the in-hospital major adverse cardiac events in the form of Univariate and multivariate analysis. P < 0.05 was set as the level of significance (P-value). Continuous data with normal distribution were expressed as mean ± standard deviation, and median (interquartile range) was used to demonstrate data with non-normal distribution.
To find the cutoff values of CAR, hs-CRP, and albumin in the prediction of short-term MACEs, the ROC curves were obtained. For comparing the area under the curve (AUC) values, the Z-test was used. P < 0.05 was taken as statistically significant two-sided analyses.
RESULTS
Overall 110 patients were enlisted in the study (89 males:29 females), among which 59 were having high CAR and 51 were having low CAR. Mean age for both high CAR and low CAR group was comparable [Table 1]. About 52.5% patients with high CAR were smokers, which were statistically significant when compared to patients with low CAR (33.3%). About 49.2% among high CAR patients were hypertensive which was statistically significant when compared to patients with low CAR. There were seven patients who had history of previous CAD and all of them had high CAR [Table 2].
| Variable | Groups | n | Mean | SD | Mean Diff | P-value |
|---|---|---|---|---|---|---|
| Age | Low CAR | 51 | 59.96 | 9.94 | 0.76 | 0.74 |
| High CAR | 59 | 59.20 | 12.40 |
| Variable | Category | Low CAR | High CAR | P-value | ||
|---|---|---|---|---|---|---|
| N | % | n | % | |||
| Gender | Males | 37 | 72.5 | 44 | 74.6 | 0.81 |
| Females | 14 | 27.5 | 15 | 25.4 | ||
| Risk Factors | Smoking | 17 | 33.3 | 31 | 52.5 | 0.04* |
| Alcohol | 18 | 35.3 | 17 | 28.8 | 0.47 | |
| T2DM | 20 | 39.2 | 33 | 55.9 | 0.08 | |
| HTN | 13 | 25.5 | 29 | 49.2 | 0.01* | |
| COPD | 1 | 2.0 | 3 | 5.1 | 0.38 | |
| Obesity | 0 | 0.0 | 4 | 6.8 | 0.06 | |
| CAD | 0 | 0.0 | 7 | 11.9 | 0.01* | |
| Hypothyroid | 0 | 0.0 | 3 | 5.1 | 0.10 | |
The percentage of major adverse cardiovascular events was in excess in patients with high CAR group than in patients with low CAR group (72.9% vs. 15.7%, P < 0.001*) [Table 3]. Males and hypertensive patients, with high CAR, had significant incidence of MACE [Table 4].
| Variable | Category | Low CAR | High CAR | P-value | ||
|---|---|---|---|---|---|---|
| n | % | n | % | |||
| In-hospital event | Cardiogenic shock | 2 | 3.9 | 13 | 22.0 | <0.001* |
| Heart failure | 6 | 11.8 | 26 | 44.1 | ||
| Reinfarction | 0 | 0.0 | 2 | 3.4 | ||
| Death | 0 | 0.0 | 2 | 3.4 | ||
| Stable | 43 | 84.3 | 16 | 27.1 | ||
| Variable | Category | MACE absent | MACE present | P-value | ||
|---|---|---|---|---|---|---|
| n | % | n | % | |||
| Gender | Males | 38 | 64.4 | 43 | 84.3 | 0.02* |
| Females | 21 | 35.6 | 8 | 15.7 | ||
| Risk Factors | Smoking | 25 | 42.4 | 22 | 43.1 | 0.94 |
| Non-smoker | 4 | 6.8 | 3 | 5.9 | 0.85 | |
| Alcohol | 16 | 27.1 | 19 | 37.3 | 0.26 | |
| T2DM | 25 | 42.4 | 28 | 54.9 | 0.19 | |
| HTN | 15 | 25.4 | 28 | 54.9 | 0.03* | |
| COPD | 2 | 3.4 | 2 | 3.9 | 0.88 | |
| Obesity | 2 | 3.4 | 2 | 3.9 | 0.88 | |
*means statistically significant P<0.05.
Multivariate and Univariate logistic regression which included statistically significant variables, including hypertension, male gender, albumin levels, hs-CRP, and CAR, revealed that CAR was found to be independently predicting MACE in patients with ACS [Table 5].
| Variable | AUC | Std. Error | 95% Conf. interval | P-value | Cut off | Sn (%) | Sp (%) | |
|---|---|---|---|---|---|---|---|---|
| Lower | Upper | |||||||
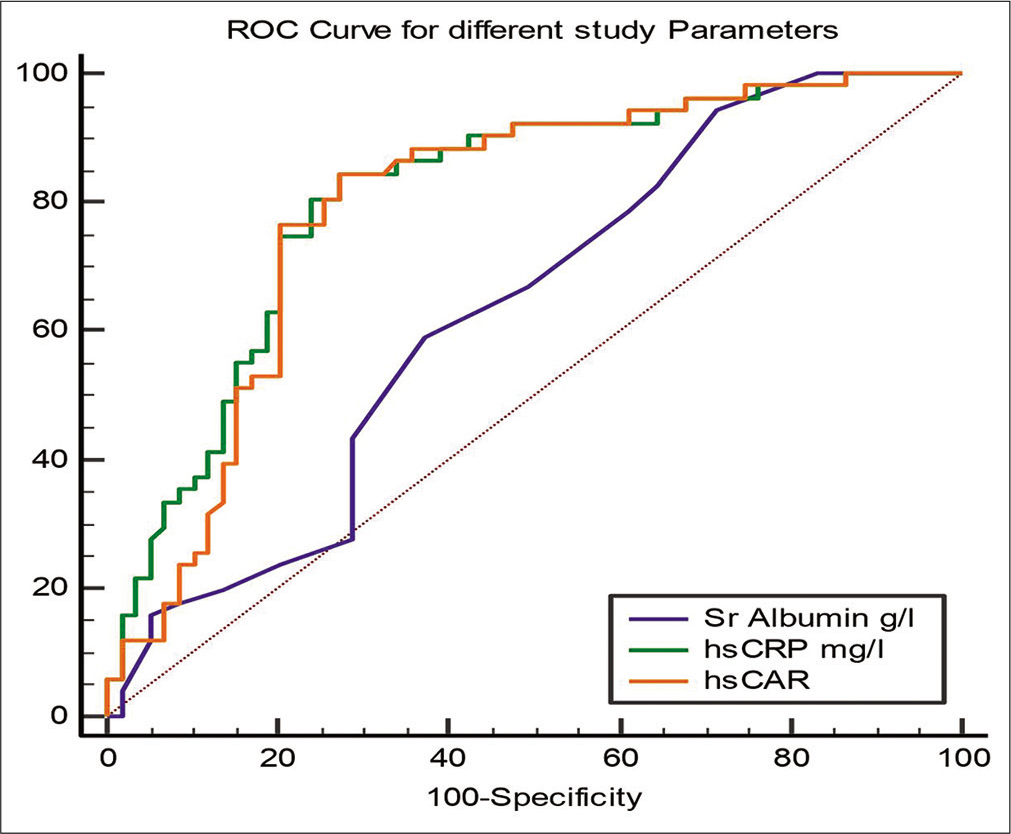
| Sr. Albumin | 0.63 | 0.05 | 0.53 | 0.72 | 0.01* | ≤42 | 94.12 | 28.81 |
| hs-CRP | 0.81 | 0.04 | 0.72 | 0.88 | <0.001* | >71.44 | 84.31 | 72.88 |
| hs-CAR | 0.79 | 0.04 | 0.71 | 0.86 | <0.001* | >1.80 | 84.31 | 72.88 |
AUC: Area under the curve
The ROC curves for CAR, hs-CRP, and albumin in predicting short-term MACEs are shown in [Figure 1]. The best cutoff value of CAR was 1.8, having 84.31% sensitivity and 72.88% specificity.
- Receiver operating curve for hs-CAR, hs-CRP, and albumin with respect to incidence of MACEs in patients with ACS.
The AUC values for CAR and hs-CRP (0.81; 95% CI: 0.72–0.88 vs. 0.79; 95% CI: 0.71–0.86 P > 0.05) did not show any significant difference. The CAR showed a significantly higher AUC value than that for albumin alone (0.79; 95% CI: 0.71–0.86 vs. 0.63; 95% CI: 0.53–0.72; P < 0.05) [Table 6].
| Parameters | Univariate analysis | Multivariate analysis | ||||||
|---|---|---|---|---|---|---|---|---|
| OR | 95.0% CI for OR | P-value | Adjusted OR | 95.0% CI for OR | P-value | |||
| Lower | Upper | Lower | Upper | |||||
| Gender, males | 2.97 | 1.18 | 7.48 | 0.02* | 2.48 | 1.05 | 7.54 | 0.04* |
| HTN | 2.46 | 1.10 | 5.55 | 0.03* | 2.00 | 1.16 | 5.83 | 0.10 |
| Sr. Albumin | 0.95 | 0.88 | 1.02 | 0.14 | 0.81 | 0.84 | 1.07 | 0.40 |
| hs-CRP | 1.20 | 1.01 | 1.30 | <0.001* | 1.04 | 0.99 | 1.09 | 0.16 |
| hs-CAR | 2.13 | 1.59 | 2.85 | <0.001* | 1.67 | 1.02 | 5.83 | 0.03* |
DISCUSSION
Rise in the hs-CRP levels is a marker of inflammation that can result in remodeling of vessels and atherosclerotic plaque rupture in coronary arteries.[15,16] A well-known study, FRISC study, included 917 patients with unstable coronary artery disease and it was seen that increased CRP levels had a strong association with the long-term sequel of death from cardiac disease.[17] In the modern interventional era, elevated hs-CRP on admission has been found to predict lesser reperfusion success.[18] As per these results, it was seen that hs-CRP has an important role in prediction in-hospital MACE in ACS patients. Moreover, increased hs-CRP levels had a direct association with CAD severity;[19] similarly, in another study, it was found that patients with triple vessel disease were having elevated levels of hs-CRP.[20] In a study by RaposeirasRoubín et al.,[21] hs-CRP was proposed to be as a predictor in-hospital MACE irrespective of higher GRACE risk score. There are some recent studies which emphasize the role of hs-CRP and serum albumin severity of CAD, namely, ACS. Yang et al. demonstrated that higher hs-CRP levels are associated with the more severe vascular lesions in ACS in recently published paper in the early 2022.[22] Furthermore, Denegri and Boriani revealed that hs-CRP is not only prognostic marker in adverse cardiovascular outcome but it also is risk factor like low-density lipoprotein cholesterol in cardiovascular atherosclerotic diseases.[23]
Keeping these studies in mind, we took hs-CRP-to-albumin ratio as a risk predictor, which is more suggestive of the same and there has been a single study till date which suggest the same. Our study revealed that hs-CRP was highly predictive of incidence of MACEs which is consistent with studies just mentioned.
After multivariate logistic regression, CAR was found to have an independent predictive power of worse ACS outcome during hospital stay. Based on the adjusted odds ratio, high CAR was associated with 67% increased risk of MACE independent of other variables.
As per the findings of our study, albumin also showed an independent association with in-hospital MACEs in ACS patients. According to few studies, low serum albumin levels have been found to have an independent association with adverse events in patients with ACS, but it still remains a debatable question whether it is so. Hartopo et al.[14] have found that low serum albumin levels do not have an association with in-hospital MACEs in patients with ACS. Chien et al.,[3] in their study, revealed that albumin had an independent predictive power for 1.5-year mortality in stable CAD patients but not in patients having heart failure. Zhu et al. published their meta-analysis in the early 2020s and demonstrated that low serum albumin level is an independent predictor of adverse outcome in patients with ACS including all-cause mortality.[24]
In our study, though low albumin was found to have an independent predictive power for MACE, but the cutoff value as per ROC was <4.2 which is higher than what was seen by Wang et al. (3.7g/dl)[25] and is also higher than 3.5 g/dl, used as a cutoff point for prognosticating worse outcome in ACS in the previous research.[14,26]
Additional studies are needed to clarify whether low serum albumin levels can independently predict adverse cardiac outcome, especially in ACS. During inflammatory processes, albumin levels are suppressed due to decreased production and increased catabolism.[27]
Apoptosis of human endothelial cells may be prevented by hypoalbuminemia.[28] As per the evidence, serum albumin has been found to have antioxidant property; it does so by binding with nitric oxide and bilirubin[29] and it also has a role in anti-platelet action and aggregation.[30,31] Furthermore, serum albumin also has been found to keep the endothelial cell membrane in a stable state and it also maintains the fluid balance across the capillary wall.[32] Keeping these things in mind, we can explain the adverse outcome in patients with lower albumin.
The hs-CAR has been found to have greater predictive power than either hs-CRP or albumin alone in prognosticating the patients with acute inflammatory states.[33] However, only fewer such studies are there, which have analyzed any correlation between CAR and MACE in ACS.
Our study revealed that CAR was very specific and sensitive marker in predicting MACE and was superior to albumin levels in predicting so, as depicted by AUC of ROC plot. CAR was almost close to hs-CRP in predicting MACE in our ACS patients with AUC slightly more with hs-CRP plot. Wang et al., in their study, also observed that CAR independently predicted MACE in ACS patients and both hs-CRP and albumin can predict adverse event.[25] In another study published in 2019, hs-CRP/prealbumin ratio was used to predict MACE, authors found that the ratio was as good in predicting MACE as the serum prealbumin alone but was superior to hs-CRP.[20] In a similar kind of study, Cagdas et al. revealed that CAR showed a greater predictive power than CRP and albumin for intermediate – high Syntax score in ACS patients, but only CRP in predicting high Syntax score II.[10]
Karabağ et al.[9] found that CAR had an independent predictive power in demonstrating no reflow in STEMI patients who were treated with primary transluminal coronary angioplasty. Furthermore, Wada et al.,[34] in their study, published that both low serum albumin and high CRP levels show a cumulative adverse effect on the risk for long-term adverse event in patients undergoing revascularization for CAD. This study confers that the CAR being a valid biomarker ratio in high inflammatory states can predict an adverse outcome.
Not more than fewer studies are there to demonstrate importance of hs-CRP to albumin ratio value in predicting worse cardiac outcome in patients with ACS. This study reiterates that CAR can act as an important biomarker tool to predict MACE and can come forth as an early predictor of any adverse event. To further enhance the validity, longer follow-ups and larger sample size are required.
Limitations
Our study being conducted for a short time period, sample size was not that large. We only assessed in hospital MACE in patients with. Longer follow-up studies are needed to demonstrate predictive power of hs-CAR in short-term and long-term adverse events in such patients.
CONCLUSION
The high sensitivity C-reactive protein to albumin ration (CAR) is an independent predictor of MACE in patients who present with ACS.
Audio summary available at
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Atherosclerosis and inflammation: Overview and updates. Clin Sci. 2018;132:1243-52.
- [CrossRef] [PubMed] [Google Scholar]
- Joint effect of carotid plaque and C-reactive protein on first-ever ischemic stroke and myocardial infarction? J Am Heart Assoc. 2018;7:e008951.
- [CrossRef] [PubMed] [Google Scholar]
- Association of low serum albumin concentration and adverse cardiovascular events in stable coronary heart disease. Int J Cardiol. 2017;241:1-5.
- [CrossRef] [PubMed] [Google Scholar]
- CDC/AHA Workshop on markers of inflammation and cardiovascular disease: Application to clinical and public health practice: Laboratory tests available to assess inflammation--performance and standardization: A background paper. Circulation. 2004;110:e572-6.
- [CrossRef] [Google Scholar]
- Prognostic value of the C-reactive protein to albumin ratio: A novel inflammation-based prognostic indicator in osteosarcoma. Onco Targets Ther. 2017;10:5255-61.
- [CrossRef] [PubMed] [Google Scholar]
- The value of preoperative Glasgow prognostic score and the C-reactive protein to albumin ratio as prognostic factors for long-term survival in athological T1N0 esophageal squamous cell carcinoma. J Cancer. 2018;9:807-15.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical significance of the C-reactive protein to albumin ratio for survival after surgery for colorectal cancer. Ann Surg Oncol. 2016;23:900-7.
- [CrossRef] [PubMed] [Google Scholar]
- The C-reactive protein/albumin ratio may predict the long-term outcome in patients with malignant pleural mesothelioma. Ann Surg Oncol. 2018;25:1471-2.
- [CrossRef] [PubMed] [Google Scholar]
- Usefulness of the C-reactive protein/albumin ratio for predicting no-reflow in ST-elevation myocardial infarction treated with primary percutaneous coronary intervention. Eur J Clin Invest. 2018;48:e12928.
- [CrossRef] [PubMed] [Google Scholar]
- Assessment of relationship between C-reactive protein to albumin ratio and coronary artery disease severity in patients with acute coronary syndrome. Angiology. 2019;70:361-8.
- [CrossRef] [PubMed] [Google Scholar]
- Relationship between C-reactive protein/albumin ratio and coronary artery disease severity in patients with stable angina pectoris. J Clin Lab Anal. 2018;32:e22457.
- [CrossRef] [PubMed] [Google Scholar]
- 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: Executive summary: A report of the American college of cardiology foundation/American heart association task force on practice guidelines. Circulation. 2013;127:529-55.
- [CrossRef] [PubMed] [Google Scholar]
- 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: A report of the American college of cardiology/American heart association task force on practice guidelines. Circulation. 2014;130:2354-94.
- [CrossRef] [PubMed] [Google Scholar]
- Low serum albumin levels and in-hospital adverse outcomes in acute coronary syndrome. Int Heart J. 2010;51:221-6.
- [CrossRef] [PubMed] [Google Scholar]
- Current concepts of the pathogenesis of the acute coronary syndromes. Circulation. 2001;104:365-72.
- [CrossRef] [PubMed] [Google Scholar]
- Relationship between coronary artery remodeling and plaque vulnerability. Circulation. 2002;105:939-43.
- [CrossRef] [PubMed] [Google Scholar]
- Markers of myocardial damage and inflammation in relation to longterm mortality in unstable coronary artery disease. FRISC Study Group. Fragmin during Instability in Coronary Artery Disease. N Engl J Med. 2000;343:1139-47.
- [CrossRef] [PubMed] [Google Scholar]
- High-sensitivity C-reactive protein and long term reperfusion success of primary percutaneous intervention in ST-elevation myocardial infarction. Int J Cardiol. 2017;248:51-6.
- [CrossRef] [PubMed] [Google Scholar]
- Long term prognostic significance of high-sensitivity C-reactive protein before and after coronary angioplasty in patients with stable angina pectoris. Am J Cardiol. 2017;99:31-5.
- [CrossRef] [PubMed] [Google Scholar]
- High sensitivity C-reactive protein to prealbumin ratio measurement as a marker of the prognosis in acute coronary syndrome. Sci Rep. 2019;9:11583.
- [CrossRef] [PubMed] [Google Scholar]
- High-sensitivity C-reactive protein is a predictor of in-hospital cardiac events in acute myocardial infarction independently of GRACE risk score. Angiology. 2012;63:30-4.
- [CrossRef] [PubMed] [Google Scholar]
- Correlation analysis of acute coronary syndrome with serum IL-18, MMP-9, hs-CRP, and plasma FIB. Biomed Res Int. 2022;2022:5984184.
- [CrossRef] [PubMed] [Google Scholar]
- High sensitivity C-reactive protein (hsCRP) and its implications in cardiovascular outcomes. Curr Pharm Des. 2021;27:263-75.
- [CrossRef] [PubMed] [Google Scholar]
- Serum albumin level for prediction of all-cause mortality in acute coronary syndrome patients: A meta-analysis. Biosci Rep. 2020;40:BSR20190881.
- [CrossRef] [PubMed] [Google Scholar]
- Prognostic efficacy of high-sensitivity C-reactive protein to albumin ratio in patients with acute coronary syndrome. Biomark Med. 2019;13:811-20.
- [CrossRef] [PubMed] [Google Scholar]
- Prognostic implications of serum albumin levels in patients with acute coronary syndromes. Am J Cardiol. 2017;119:951-8.
- [CrossRef] [PubMed] [Google Scholar]
- Acute-phase proteins and other systemic responses to inflammation. N Engl J Med. 1999;340:448-54.
- [CrossRef] [PubMed] [Google Scholar]
- Serum albumin is a specific inhibitor of apoptosis in human endothelial cells. J Cell Sci. 1996;109:2571-80.
- [CrossRef] [PubMed] [Google Scholar]
- Redox properties of serum albumin. Biochim Biophys Acta. 2013;1830:5465-72.
- [CrossRef] [PubMed] [Google Scholar]
- Anticoagulant action of low, physiologic, and high albumin levels in whole blood. PLoS One. 2017;12:e0182997.
- [CrossRef] [PubMed] [Google Scholar]
- Histone induced platelet aggregation is inhibited by normal albumin. Thromb Res. 2013;132:69-76.
- [CrossRef] [PubMed] [Google Scholar]
- Human serum albumin in the clinical syndrome of heart failure. J Card Fail. 2011;17:451-8.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of a modified early warning system for acute medical admissions and comparison with C-reactive protein/albumin ratio as a predictor of patient outcome. Clin Med. 2009;9:30-3.
- [CrossRef] [PubMed] [Google Scholar]
- Independent and combined effects of serum albumin and C-reactive protein on long-term outcomes of patients undergoing percutaneous coronary intervention. Circ J. 2017;81:1293-300.
- [CrossRef] [PubMed] [Google Scholar]







