Translate this page into:
Risk Factors in Young Female Patient with Polycystic Ovary Syndrome presenting with Acute Myocardial Infarction
Vivek Vilas Manade, MD Department of Cardiology Dr. D. Y. Patil Medical College, Pimpri, Pune 411018, Maharashtra India dr.vivek.manade7@gmail.com
This article was originally published by Thieme Medical and Scientific Publishers Pvt. Ltd. and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Abstract
Polycystic ovary syndrome (PCOS) is known to cause weight gain, nonspecific inflammation, dyslipidemia, and coronary artery disease (CAD).
Oral contraceptives (OCs) also cause venous and arterial thromboembolism, cerebrovascular accidents (CVA), and acute coronary syndrome (ACS). Myocardial infarction (MI) is comparatively less common in patients taking newer generation OCs, particularly if risk factors for CAD are absent, but the risk for venous thromboembolism is still high.
Therefore, OCs should be considered in properly selected patients after weighing risks and benefits.
Keywords
acute myocardial infarction
low-dose oral contraceptives
young female
Introduction
Polycystic ovary syndrome (PCOS) is associated with acute coronary syndrome (ACS), particularly acute myocardial infarction (AMI) secondary to obesity, low-grade inflammation, dyslipidemia, and endothelial dysfunction. This risk of AMI further increases with the presence of traditional risk factors for MI.
We are presenting a case of a young female on oral contraceptives (OCs) for PCOS with total thrombotic occlusion of left anterior descending (LAD) artery causing AMI with multiple risk factors.
Case Report
A 22-year-old female residing at Pimpri-Pune, 2nd year M.A. student, presented with chest pain for a period of 8 to 9 hours. The patient was apparently stable in the morning, but then she developed a sudden onset of chest pain while having her lunch at approximately 2:30 PM. The pain was burning in nature, lasting for a few minutes, radiating to the epigastric region, and not being relieved by taking antacids and pain killers. It was not associated with cough, expectoration, dyspnea, palpitations, syncope, fever, trauma, and nausea or vomiting. The intensity of chest pain increased at approximately 3:00 PM for which she came to Dr. D. Y. Patil Hospital, Pimpri, Pune at about 4:00 pm She was hemodynamically stable. Her ECG on arrival (Fig. 1) and subsequent serial ECGs were normal. Her CPK-MB was 28 and Trop-T negative. She was advised hospitalization but she refused to stay.
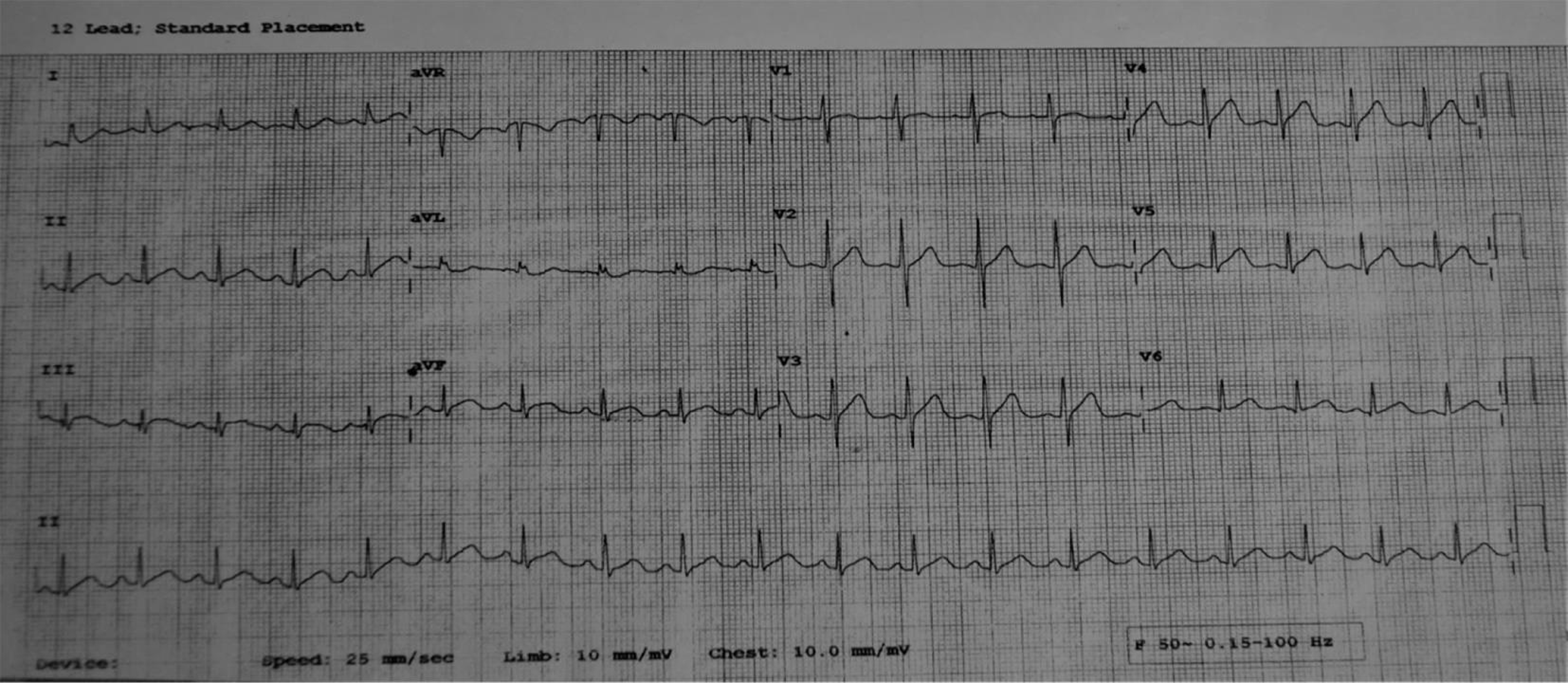
-
Fig. 1 1st ECG on presentation.
Fig. 1 1st ECG on presentation.
At about 9:00 PM , she developed severe retrosternal chest pain associated with profuse sweating and giddiness. The pain was radiating to the back, neck, and left arm, and persistent. She was brought by her friends to the hospital at approximately 11:00 PM. Her ECG was showing extensive anterior wall MI (Fig. 2).
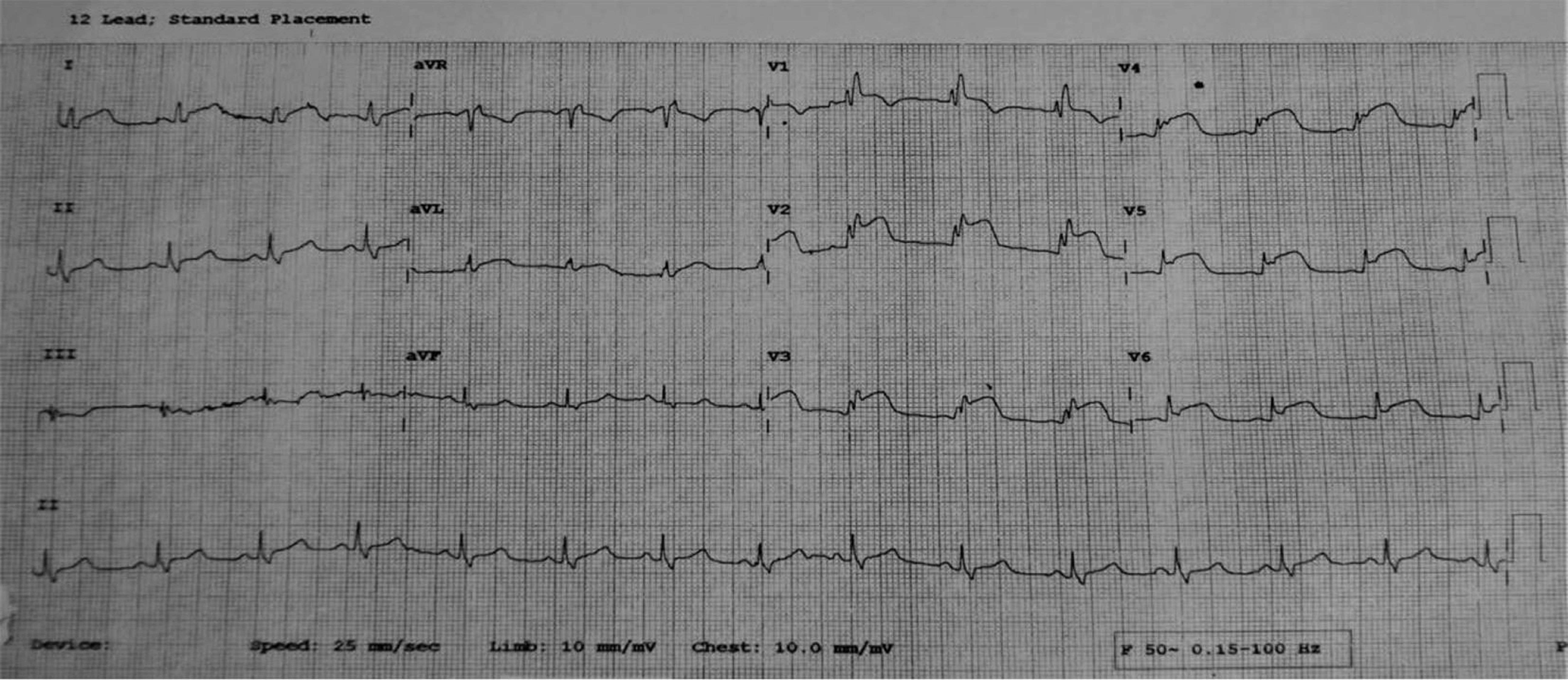
-
Fig. 2 2nd ECG on presentation: AWMI. Abbreviation: AWMI, anterior wall myocardial infarction.
Fig. 2 2nd ECG on presentation: AWMI. Abbreviation: AWMI, anterior wall myocardial infarction.
Her ECHO revealed apical, apico-septal, midseptal, and apico-anterior segments were akinetic. EF: 35%
She was K/C/O PCOS for 3 to 4 years and on regular treatment. She was taking tablet DEVIRY 10 mg (medroxyprogesterone) one tablet daily for 3 to 4 years continuously and tablet ORAL-L (0.15 mg levonorgestrel and 30 μg Ethinyl estradiol) since the last 1 month once daily along with some Ayurvedic medications.
Her parents were afflicted with hypertension (HTN), and her mother had h/o two spontaneous abortions, with no h/o any major cardiac events in family members.
She experienced emotional stress regarding her personal issues for the last 1 year.
She was hemodynamically stable. Her general and systemic examination was normal, except obesity with body mass index (BMI) of 32 Kg/m2.
Primary treatment was given and the patient was shifted to the Cath-laboratory for primary percutaneous coronary intervention (PCI) (radial approach) with door to balloon time being < 90 minutes (80 minutes).
Coronary angiography (CAG) showed total thrombotic occlusion of LAD from midsegment, thrombotic occlusion of 1st septal and diagonal branch, with some thrombus also noted in ostioproximal LAD. (Fig. 3) Left circumflex (LCx) artery was normal, ramus was normal, and right coronary artery (RCA) was dominant and normal.
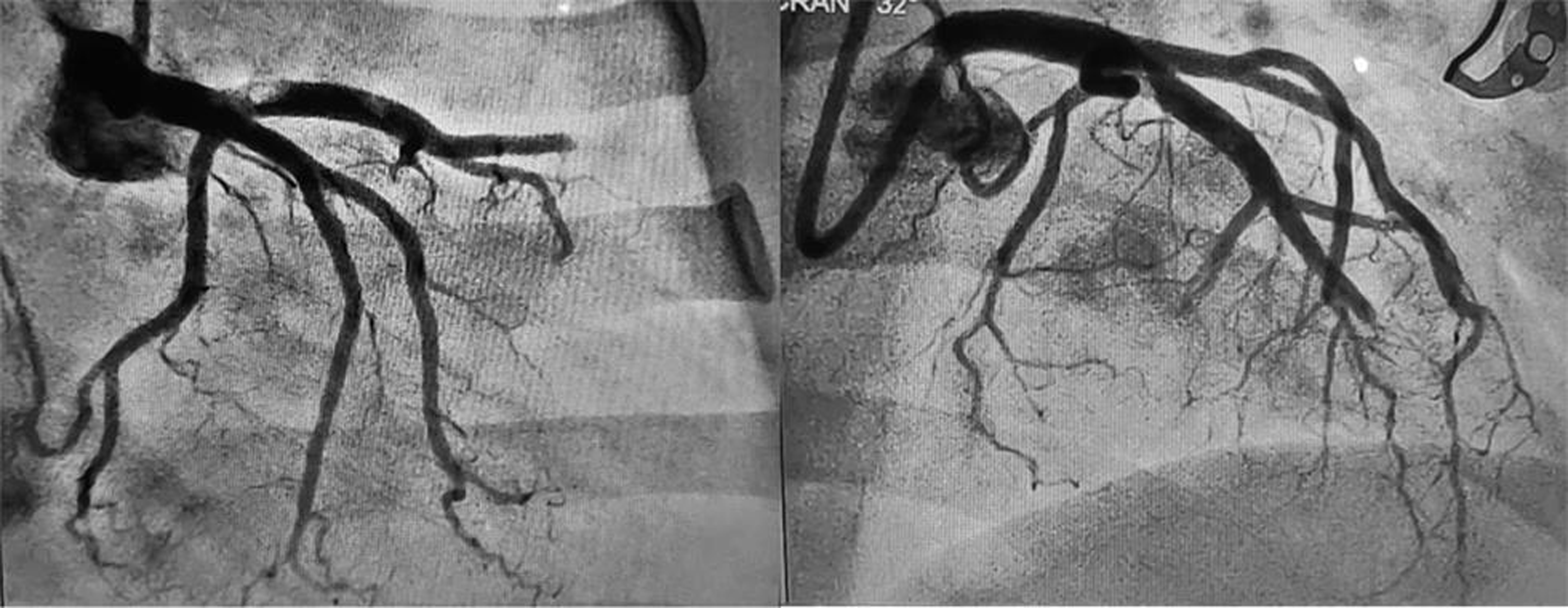
-
Fig. 3 Total thrombotic occlusion of LAD artery in RAO caudal and LAO cranial views. Abbreviations: LAD, left anterior descending; LAO, left anterior oblique; RAO, right anterior oblique.
Fig. 3 Total thrombotic occlusion of LAD artery in RAO caudal and LAO cranial views. Abbreviations: LAD, left anterior descending; LAO, left anterior oblique; RAO, right anterior oblique.
Manual aspiration of thrombus was done (Fig. 4 ), and stenting was deferred as stenosis was less than 50%. Fig. 5 represent results of postthrombosuction.
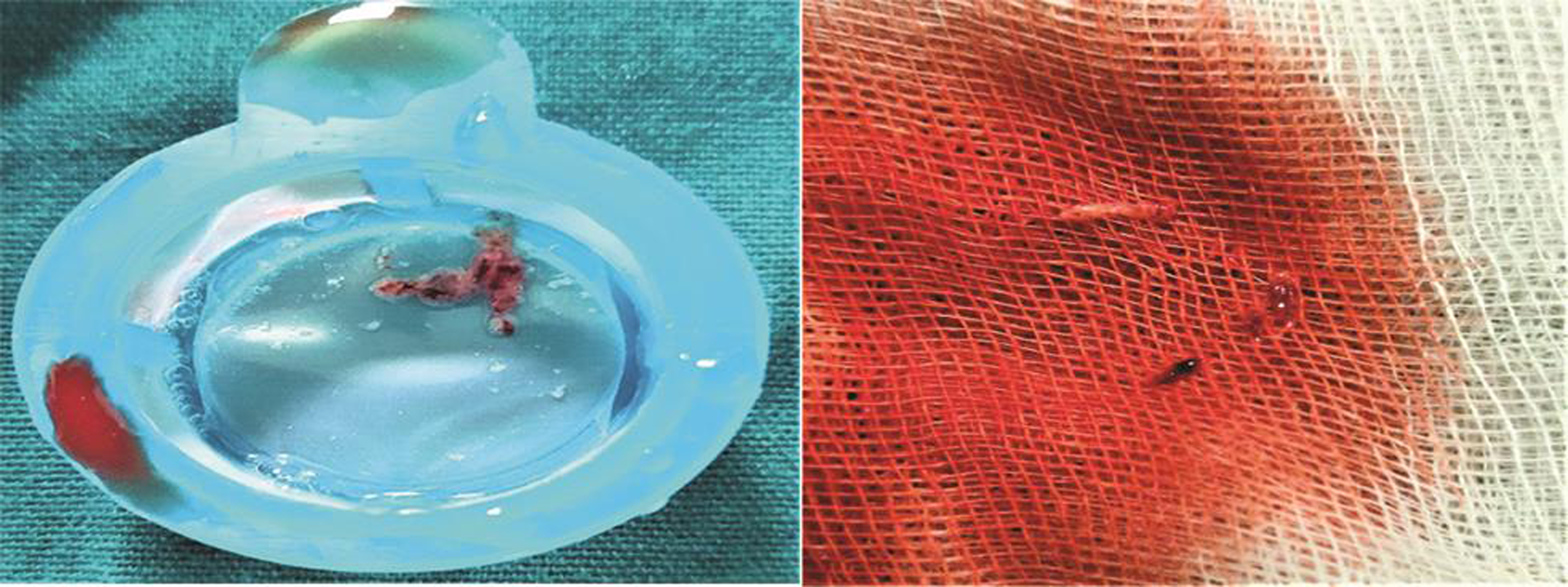
-
Fig. 4 Aspirated thrombus.
Fig. 4 Aspirated thrombus.
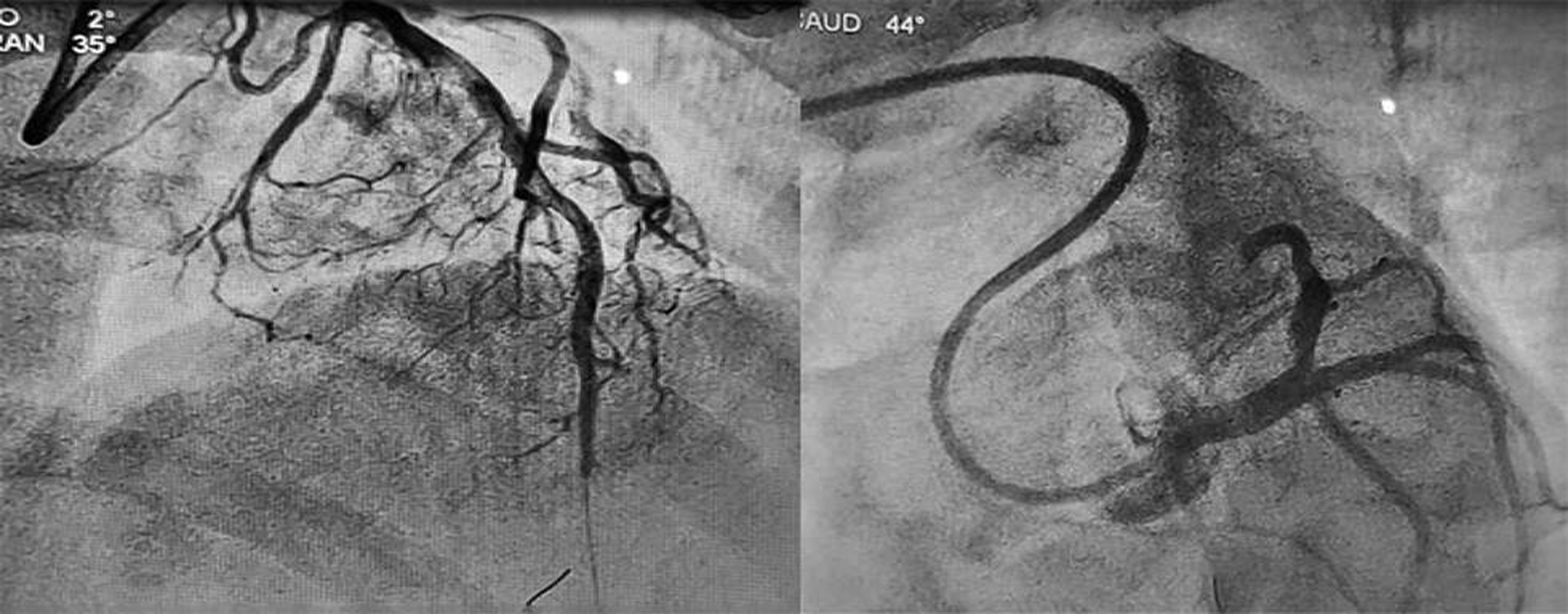
-
Fig. 5 Postthrombosuction: AP cranial and LAO caudal views showing decreased thrombotic burden with insignificant residual stenosis in LAD. Abbreviations: LAD, left anterior descending; LAO, left anterior oblique.
Fig. 5 Postthrombosuction: AP cranial and LAO caudal views showing decreased thrombotic burden with insignificant residual stenosis in LAD. Abbreviations: LAD, left anterior descending; LAO, left anterior oblique.
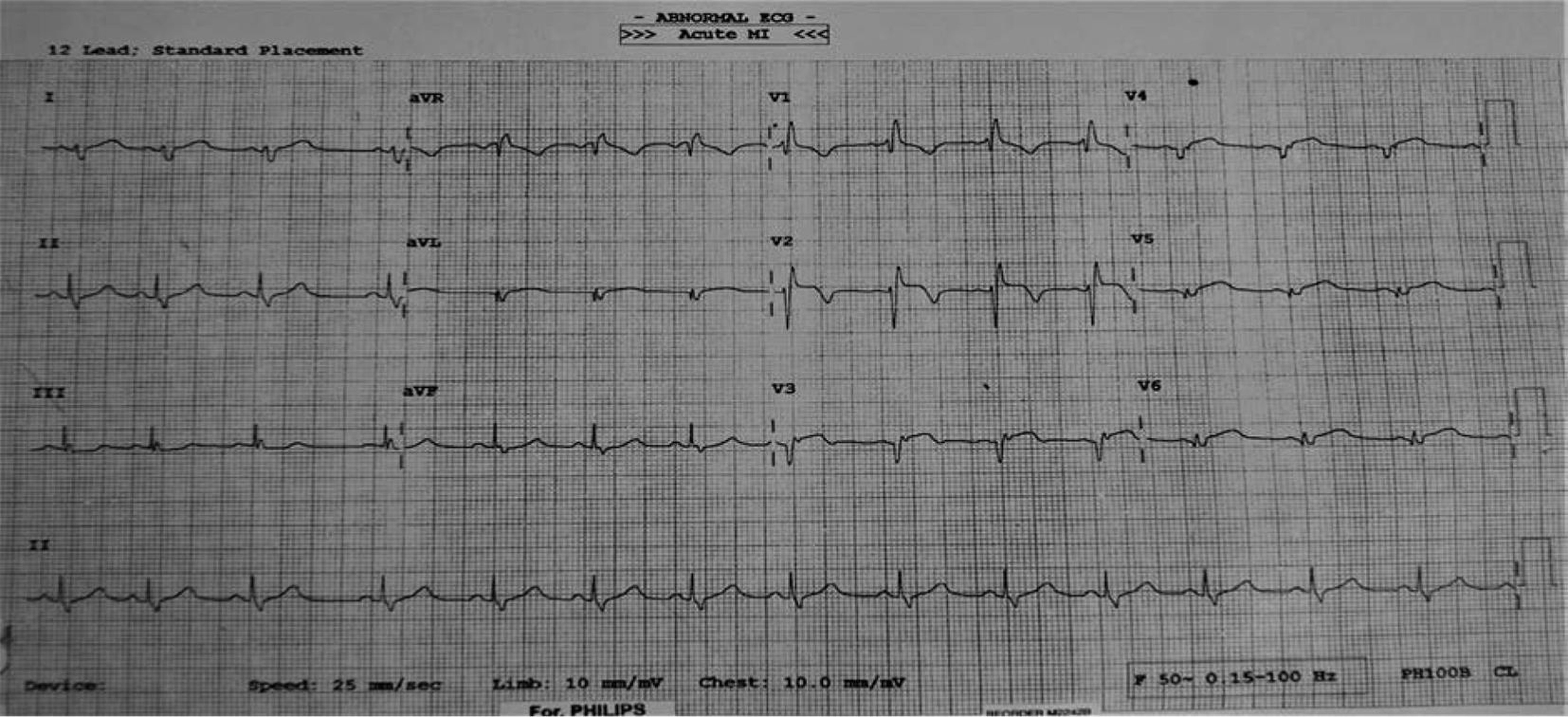
Post-PCI, patient was hemodynamically stable and shifted to CCU for further care. Post-PCI ECG revealed almost isoelectric ST segment without any chest discomfort (Fig. 6). Along with routine post-PCI regimen, patient was started on Tirofiban infusion in standard dose for 24 hours, and check shoot angiography after 24 hours revealed TIMI grade 3 flow with residual insignificant stenosis. (Fig. 7)
-
Fig. 6 Post-PCI ECG: Almost isoelectric ST segment in anterior leads.
Fig. 6 Post-PCI ECG: Almost isoelectric ST segment in anterior leads.
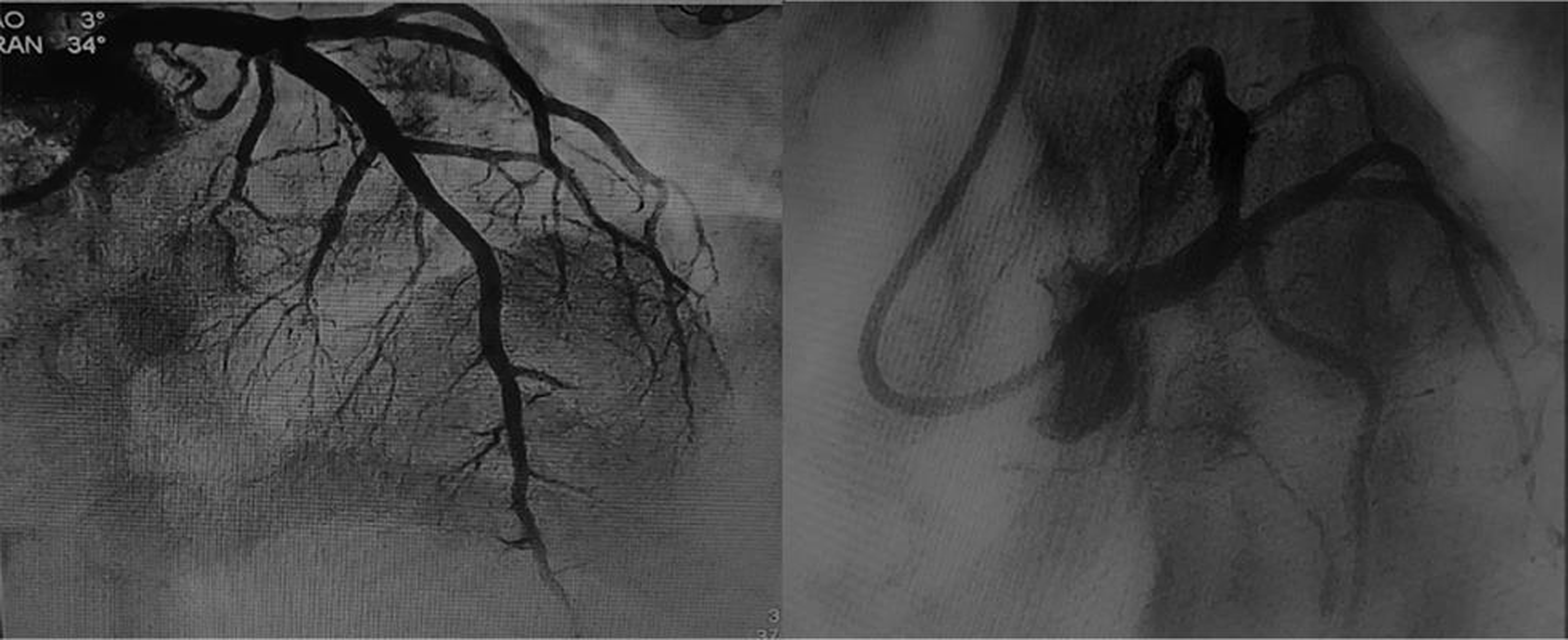
-
Fig. 7 Post-PCI final result (after 24 hours of Tirofiban infusion), AP cranial and LAO caudal views showing TIMI grade 3 flow with insignificant residual stenosis in LAD. Abbreviations: LAD, left anterior descending; LAO, left anterior oblique; TIMI, thrombolysis in myocardial infarction.
Fig. 7 Post-PCI final result (after 24 hours of Tirofiban infusion), AP cranial and LAO caudal views showing TIMI grade 3 flow with insignificant residual stenosis in LAD. Abbreviations: LAD, left anterior descending; LAO, left anterior oblique; TIMI, thrombolysis in myocardial infarction.
The patient’s hemogram was normal, except WBC: 17,200/microL. Her CPK-MB was 258 and TROP- I was 47,605. Her lipid profile, TFT, and CXR were normal. Serum B12 was 148 pg/L (lower limit of normal range), homocystein was 57 microgram/L (elevated > 15), folic acid was 4.3 (normal), and antinuclear antibody (ANA) blot assay: Negative and coagulation profile revealed isolated protein C deficiency.
Patient was hemodynamically stable at the time of discharge. The patient was regularly attending follow-up and doing well on medications. Follow-up ECHO showed EF 60%
The patient was having anterior wall STEMI with risk factors, PCOS, OC pills, obesity (BMI > 32 kg/m2), hyperhomocysteinemia, isolated protein C deficiency, emotional stress, and endothelial dysfunction.
Discussion
Risk factors for MI in young patients include traditional risk factors for atherosclerosis and other factors such as F/H/O premature coronary artery disease (CAD), coronary embolism, coronary spasm, coronary dissection, coronary aneurysm, coronary vasculitis, collagen vascular diseases, hypercoagulability states, drugs, and stress.
Unique and novel risk factors that occur more commonly or exclusively in women are preeclampsia, eclampsia, pregnancy-induced hypertension (PIH), gestational diabetes, systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), and psychosocial: anxiety, depression, job-marital stress.1 2 3
PCOS is associated with increased risk of DM, HTN, and dyslipidemia causing endothelial dysfunction.4 5 6 Hyperandrogenism (free testosterone) in PCOS is associated with systolic blood pressure7 and abnormal lipid metabolism.8
If traditional risk factors are present in women taking OCs, then underlying atherosclerosis process is augmented, subsequently increasing the risk of ACS at a younger age.9
Estrogen-containing pills are more prone for the development of thrombotic events, both arterial and venous, than progesterone-containing pills. However, there are some studies showing increased thrombotic risk with the use of progesterone-only pills for longer duration of time (in years) by unknown mechanisms. In our case, the patient was taking tablet DEVIRY 10 mg (medroxyprogesterone) for 3 to 4 years.
Even a low-dose OC for a shorter period of time even without any risk factors for CAD can cause AMI but incidence is rare.10 Therefore, treating physicians as well as patients on OCs must be aware of signs, symptoms and risk factors of CAD. Care must be taken to avoid, at least, modifiable risk factors for MI. In our case, the patient was taking tablet ORAL-L (0.15/0.03 mg) since the last 1 month.
Isolated protein C deficiency can occur in patients taking OCPs for a longer duration of time.11
Serum homocysteine can be elevated in some patients of PCOS and also be associated with increased risk of vascular thromboembolism.12
Endothelium is the inner most lining of blood vessel (Tunica intima) by monolayer of endothelial cells. It lines the entire circulatory system: from heart to capillaries.13 The functions of endothelium are as follows: acts as a physical barrier between the blood and vessel wall, transports nutrients between blood and tissues, and serves anti-inflammatory, antithrombotic and vasoregulatory purposes. Normal endothelial function is characterized by a fine balance between vasoconstrictors and vasodilators, inflammatory and anti-inflammatory factors, and thrombotic and antithrombotic factors.14 15 16
Endothelial dysfunction occurs as a result of decreased vasodilators (NO), increased vasoconstrictors, increased oxidative damage, increased inflammatory factors, increased thrombotic factors, and increased atherosclerotic factors, ultimately causing thrombotic occlusion of vessel following atherosclerotic plaque rupture/ erosion.14 15 16
Conclusion
Incidence of CAD is increasing among those < 40 years. Detailed history, thorough clinical examination along with relevant investigations helps us to detect early diagnosis of AMI. But above all, suspicion regarding ACS is the most important factor in managing these cases. Most of the young patients have atherosclerotic plaque erosion f/b thrombotic occlusion of culprit vessel and have a tendency toward complete recanalization without need of coronary stenting. Along with optimal medical treatment, identification and treatment for modifiable risk factors is also mandatory.
In symptomatic women with angina, despite the absence of obstructive stenosis on CAG, one must consider and further investigate coronary microvascular dysfunction, endothelial dysfunction, microvascular spasm, spontaneous coronary artery dissection, and other nonvascular myocardial causes.
Conflict of Interest
None.
References
- Effectiveness-based guidelines for the prevention of cardiovascular disease in women–2011 update: a guideline from the american heart association. Circulation. 2011;123(11):1243-1262.
- [Google Scholar]
- Sex differences in mental stress-induced myocardial ischemia: are women from venus? J Am Coll Cardiol. 2014;64(16):1679-1680.
- [Google Scholar]
- Sex and age differences in the association of depression with obstructive coronary artery disease and adverse cardiovascular events. J Am Heart Assoc. 2014;3(03):e000741.
- [Google Scholar]
- Epidemiology and adverse cardiovascular risk profile of diagnosed polycystic ovary syndrome. J Clin Endocrinol Metab. 2006;91(04):1357-1363.
- [Google Scholar]
- Adverse lipid and coronary heart disease risk profiles in young women with polycystic ovary syndrome: results of a case-control study. J Clin Epidemiol. 1998;51(05):415-422.
- [Google Scholar]
- Endothelial dysfunction in young women with polycystic ovary syndrome: relationship with insulin resistance and low-grade chronic inflammation. J Clin Endocrinol Metab. 2004;89(11):5592-5596.
- [Google Scholar]
- Relationship between serum sex hormones and coronary artery disease in postmenopausal women. Arterioscler Thromb Vasc Biol. 1997;17(04):695-701.
- [Google Scholar]
- Coronary heart disease risk factors in women with polycystic ovary syndrome. Arterioscler Thromb Vasc Biol. 1995;15(07):821-826.
- [Google Scholar]
- Low-dose oral contraceptive-induced acute myocardial infarction. Eur J Contracept Reprod Health Care. 2016;21(06):499-501.
- [Google Scholar]
- Isolated protein S deficiency presenting as catastrophic systemic arterial and subsequently venous thrombosis. Australas Med J. 2012;5(08):424-428.
- [Google Scholar]
- Diastolic dysfunction and increased serum homocysteine concentrations may contribute to increased cardiovascular risk in patients with polycystic ovary syndrome. Fertil Steril. 2001;76(03):511-516.
- [Google Scholar]
- 2007. p. :1-14. The vascular endothelium: structure and function. In: Fitridge R, Thompson M, eds. Mechanisms of Vascular Disease A TEXTBOOK FOR Vascular Surgeons. 1st ed. Cambridge University Press
- The endothelium: physiological functions and role in microcirculatory failure during severe sepsis. Intensive Care Med (e-pub ahead of print).
- [CrossRef] [Google Scholar]







