Translate this page into:
Hypertension in Women: The Current Understanding and Future Goals
*Corresponding author: Prerna Goyal, Department of Medicine, RG Stone and Super-speciality Hospital, Ludhiana, Punjab, India. drprerna156@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Goyal P, Chhabra ST, Jyotsna M. Hypertension in women: The current understanding and future goals. Indian J Cardiovasc Dis Women 2022;7:106-16.
Abstract
Hypertension is one of the major contributing risk factor of cardiovascular diseases. Despite enormous advances in the preventive cardiology, hypertension remains the leading cause of death and disability in women. The article aims to focus on the gender specific differences in hypertension, and existing gaps in the current understanding of high blood pressure (BP) in women. Apart from epidemiological differences, hypertension in men and women has distinct pathophysiological mechanisms, impact on cardiovascular system, awareness and control. Prevalence of hypertension is higher in men than women till menopause; following this the prevalence rises steeply in women, and exceeds that of men above 75 years of age. Women with their estrogenic environment are relatively protected from high BP as their hormonal/chromosomal profile govern expression of alternate renin angiotensin axis (RAS) pathway and anti-inflammatory, vasodilatory, anti-proliferative immune cells whereas in males, classical RAS driven inflammatory, pro-hypertensive and proliferative milieu confers higher risk of hypertension. Thus, immunotherapy can have a potential therapeutic role in the treatment of hypertension in future. Cardiovascular consequences of high BP are worse in women than men in majority of trials. Women are now getting more aware of hypertension but the control of BP still remains poorer than men, especially in older age group. There are some noteworthy pharmacokinetic and pharmacogenomics gender differences in response to various antihypertensive drugs, which can be taken into consideration while choosing a particular class of drugs in female population. Standard treatment guidelines recommend same BP targets and management strategies in both the genders, but the trials so far have not been designed in a way to draw women specific conclusions on optimal cut-offs for diagnosis and treatment of BP due to under representation of women in majority of trials. More women centered analysis in future hypertension research projects can provide better scientific insights in various clinical aspects of hypertension.
Keywords
Gender
Cardiovascular
Blood pressure
INTRODUCTION
Cardiovascular (CV) disease is one of the leading causes of death and disability in both males and females. Women in postmenopausal age constitute the highest population burden of sudden cardiac death (13% of all deaths and 50% of all coronary deaths). Of all the modifiable risk factors for all cause death and disability in women, hypertension stands at the foremost position in the list.[1] The average life expectancy of women with hypertension has been reported to be approximately 5 years shorter than women with normal blood pressure (BP) at 50 years of age. Women’s Health Initiative in its report also singled out high BP as a prime independent risk factor for sudden cardiac death in post-menopausal women.[2] As there is an exponential relation between high BP and CV mortality, even a smaller attempt to control BP in females can make a huge impact in lowering down the death rate in women.[3] There have been tremendous advances in the current understanding of gender differences in hypertension but an extensive review of literature calls for more research to explore further the intricacies of hypertension in women.[4] This article aims to present a focused review on various issues related to hypertension in women, highlighting the gender dimorphism in the epidemiology, pathophysiology, CV effects, and pharmacological response to various drugs of hypertension.
SEX VERSUS GENDER: AN OVERLOOKED TRAIT
It is essential to understand the differences between these two synonymously but distinct terms – gender and sex. As defined by the Institute of Medicine Committee on Understanding the Biology of Sex and Gender Differences, sex is a chromosomal driven biological and anatomical identity of an individual assigned at birth as male or female whereas gender is a social and behavioral construct based on individual’s self-revelation as man/boy or woman/ girl.[5] Gender of a person may or may not match with the sex assigned at birth. Knowing the gender or sex identity of a person is important as our basic conceptual science is based on sexual phenotype whereas much of data collection or research activities to study the impact of a disease process or therapy are based on self-representation of gender at population level. For this reason, the term “sex” is used while referring to basic scientific developments examining male versus female phenotype and the term “gender” refers to clinical studies examining man versus woman identification.
EPIDEMIOLOGICAL DIFFERENCES IN HYPERTENSION: DOES GENDER MATTER?
There has been a remarkable increase in the overall prevalence of hypertension among all age groups in both men and women following the American College of Cardiology/ American Heart Association (ACC/AHA) 2017 Hypertension guidelines. Global figures estimate that nearly 1.13 billion people are hypertensive, two-third of which live in low- and middle-income countries.[6] About one out of four men and one out of five women have high BP. In India also, hypertension prevalence is increasing like an epidemic with a recorded 3-fold increase from 2004–2005 to 2011–2012,[7] accounting for 5.1% of total deaths and 15% of CV deaths.[8]
Age-related gender differences
It has now been well established from the global statistics that men are more prone to hypertension than women in premenopausal age group; afterward, the prevalence rises at a much rapid rate in women, and subsequently overshoots in elderly postmenopausal women.[9] National Health and Nutrition Examination Survey BP data from 2011 to 2014 using the Joint National Committee (JNC) 7/8 BP guidelines report comparable prevalence in both the genders between 20–44 and 55–74 years of age, men being more affected in the age group 45–54 and women exceeding men above 75 years of age.[10]
Interestingly, the age-related gender differences in hypertension are found to be exaggerated after application of ACC/AHA 2017 hypertension criteria of BP. Men now have a higher prevalence of hypertension starting from 20 years of age till 65, the prevalence gap then narrows between 65–74 and reverses after 75 years of age. This age-related increase in male prevalence in majority of age groups below 65 years is an indirect evidence that historically there were more men with prehypertension than women as per JNC 7 criteria. A large meta-analysis including 120,605 men and 130,136 women from 13 countries further supported the notion of high-pooled prevalence of prehypertension among men (40%) versus women (33%).[11]
Demographical variables in relation to hypertension in Indian population
The large database from the National Family Health Survey 2015–16 (NFHS-4) provided the following reliable information on sex/gender specific differences in the prevalence of hypertension among the Indian men and women; stratified by socioeconomic status, geographical distribution, cultural, behavioral, and lifestyle characteristics.[12]
Indian data also mimic the global trends of overall high prevalence in men (16.32%) than women (11.56%).
Hypertension prevalence rises with increasing age, older adults (40–54 years) having the highest and almost double the prevalence as compared to younger individuals (25–39 years).
State-wise distribution showed that residents of North, Northeast, and Southern states except Kerala have a relatively higher prevalence of hypertension.
The prevalence of hypertension is more among men compared to women in majority of states except Delhi where the prevalence among women is higher than men (7.18% vs. 5.22%, respectively).
Punjab reported the highest prevalence of hypertension among men (25.9%) and Sikkim among women (18.8%).
The urban residents and people from poor wealth quintile and married adults are more hypertensive than their respective counterparts.
Daily consumption of alcohol and non-vegetarian diet is strongly associated with high BP.
-
Gender dimorphism-
Older age women, with low level of education, non-working environment, history of consumption of alcohol, presence of diabetes, and Muslim religion had higher prevalence of hypertension than men.
Married, non-vegetarians, and highly educated men engaged in professional activities (managerial/ sales/administrative) have more odds of having hypertension than women.
HYPERTENSION AND ITS CV IMPACT IN WOMEN
The direct relationship between hypertension and CV disease has been established in various trials since 1960s. Every 20 mm Hg increase in systolic BP or 10 mm Hg increase in diastolic BP almost doubles the risk of CV diseases.[13] In addition, 24-h ambulatory and conventional daytime and nighttime systolic BP recordings are found to be significant predictors of CV events. Ambulatory BP monitoring has also been suggested to be the preferred method of recording BP in high-risk pregnancies for early detection of adverse maternofetal events in a study by Vavilala et al.[14]
However, the impact of high BP is different for men and women.[15] Hypertensive women have been found to have more of non-traditional CV risk factors such as central obesity and kidney disease as compared to hypertensive men with more of traditional risk factors such as smoking and dyslipidemia.[16] For a 10 mmHg increase in systolic BP, the CV risk in women rises by 25% compared to only 15% rise in men.[17] Besides this, perimenopausal and menopausal women exhibit more variation in 24-h ambulatory BP measurements[18] and a non-dipping pattern of BP,[19] which may confer poor cardiovascular outcomes and target organ damage in older women.[20] A prospective study by Hermida et al. including 3344 participants (48.6% women) demonstrated a stronger relationship between high ambulatory BP and CV risk in women than in men. They also reported optimal BP threshold for CV protection as daytime BP of 135/85 mm Hg in men and 125/80 mmHg in women; and night time BP of 120/70 mm Hg in men and 110/65 mmHg in women.[21]
Hypertension leading to deleterious end organ consequences such as left ventricular hypertrophy, heart failure with preserved ejection fraction, diastolic dysfunction, diabetes, and chronic kidney disease are more common in women than men.[22] A prospective cohort study of participants in Reasons for Geographic and Racial Disparities in Stroke trial found a strong association between increasing hypertension severity and incident ischemic stroke in women compared with men.[23] Another community-based cohort study found an association between cardioankle vascular index (CAVI); a marker of subclinical atherosclerosis and aortic stiffness and BP category in men and women.[24] They found that CAVI increased as the stage of hypertension progressed in males but remained high in all stages of hypertension in females suggesting that optimal management strategy should focus not only on age, comorbidities but also gender. A research paper by Nethi et al. reported a rise in carotid-femoral pulse wave velocity in hypertensive women in parallel with the grade of hypertension, confirming the presence of vascular remodeling and increase in arterial stiffness which, in turn, marks the adverse CV consequences in women.[25]
Contrary to all these findings, a meta-analysis (5018 participants) did not find any significant gender differences between conventional home BP measurements and 10-year CV risk.[26]
One of the reasons for this discrepancy in CV outcomes could be difference in the methodology used to measure BP (ambulatory/conventional BP measurement) or the time of day when BP was recorded. Regardless of the reason, there is sufficient evidence that signify a relative higher CV risk in hypertensive females as compared to hypertensive males and makes sense to raise an important fundamental question – “should the optimal outcome based BP target be the same in females as males?”
Possible explanations for the higher CV risk in hypertensive female cohort
Difference in biologic, behavioral, or hormonal characteristics of women that strongly link hypertension to vascular dysfunction.
Gender-specific synergism between hypertension and other CV risk factors that contribute to poorer outcomes in women.
Differences in the pharmacological treatment and poorer therapeutic adherence can be the other possibility of increased CV risk in women.
PATHOPHYSIOLOGY OF HYPERTENSION UNIQUE TO WOMEN
A woman in her life span encounters a varied spectrum of biological transformation; from young adulthood through reproductive age to menopause and old age when each phase has its own implications in onset of hypertension [Figure 1]. Hypertensive disorders of pregnancy, reproductive assisted technologies, and hormonal oral contraceptives/ menopausal hormonal replacement therapies have their own pathophysiological mechanisms of hypertension in women population.

- Hypertension in women across different stages of life span.
Passing over these women specific attributes of hypertension, estrogens in females play an indispensible protective role in maintaining the vascular hemostasis. Estrogens up-regulate nitric oxide (NO) pathway by increasing NO synthetase activity, inhibit endothelin production, and down-regulate the renin angiotensin system (RAS) by decreasing renin and angiotensin converting enzyme activity (ACE) activity and Angiotensin II (Ang II) production.[27-29] Estrogens also reduce the oxidative stress and inflammation through decreasing the generation of reactive oxygen species, increasing the antioxidant production, and inhibition of pro-fibrotic genes. This explains well the increased risk of hypertension seen in post-menopausal hypoestrogenic women. The hemostatic effects of estrogens are also supported by the fact that younger women with polycystic ovarian disorder or infertility or premature ovarian insufficiency due to any kind of estrogen imbalance have an increased risk of developing hypertension.[30]
The two major systems that contribute to sex differences in pathophysiology of hypertension are:
RAS
Immune System
Sex and gender differences in RAS expression
Renin angiotensin axis in our body works in two contrasting pathways – classical and non-classical/ alternative. Angiotensinogen from liver is converted into angiotensin I (Ang I) by enzyme renin. ACE 1 further cleaves Ang I to Ang II, which then bind to one of the two angiotensin receptors [ATR].[31] The receptor expression has been found to be primarily under the regulatory control of estrogen and chromosome complement. The basic scientific studies confirm that males have biologically greater expression and physiological responses to activation of the classical RAS pathway (includes Ang II, Ang II type 1 receptor AT1R stimulation, and ACE), whereas females have greater expression and physiological responses to activation of the non-classical RAS pathway (includes Ang 1–7, Ang II Type 2 receptor AT2R, Mas receptor, and ACE2) [Figure 2].
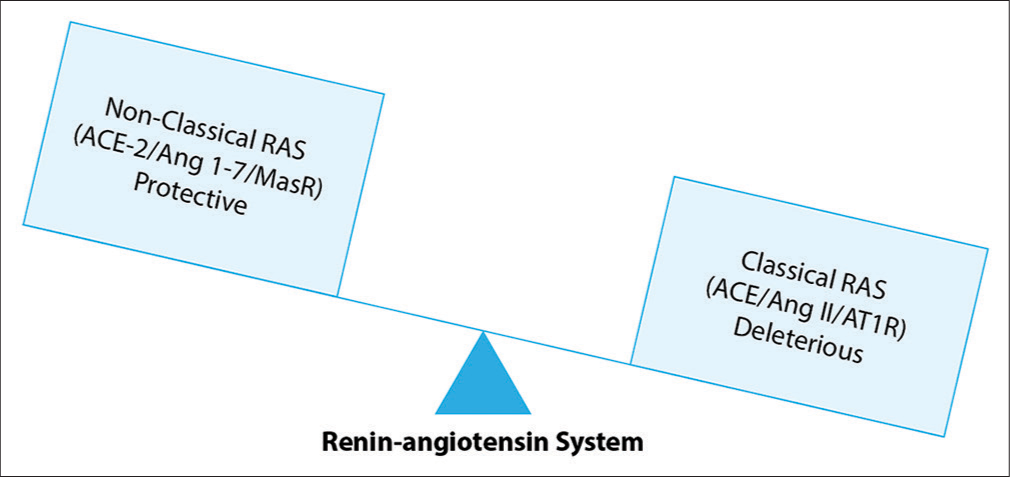
- Physiological effects of key peptides of classical versus non-classical RAS.
This means in males, Ang II predominantly binds to AT1R which leads to vasoconstriction, renal tubular sodium reabsorption, and rise in BP through activation of classical RAS pathway. In contrast, in females, Ang II binds to AT2R, which triggers vasodilation and natriuresis through activation of alternate RAS pathway. Another component of non-classical/alternate RAS in females is generation of vasodilatory peptide Angiotensin (1–7) peptide from Ang II by the action of enzyme ACE2. Angiotensin (1–7) binds to the Mas receptor and promotes vasodilation and natriuresis.[32] It also attenuates the hypertensive effects of Ang II [Figure 3].

- Simplified representation of major angiotensin pathways with their respective peptides, receptors, and effects.
Apart from these sex divergent effects of angiotensin receptor on hypertension, AT2R stimulation creates an anti-inflammatory milieu in females whereas AT1R stimulation promotes inflammation, proliferation, and angiogenesis in males. This sex specific expression of Ang II receptor in kidneys and vasculature has been validated in spontaneously hypertensive male and female rats models.[33,34]
Role of immune cells in sex differences in hypertension
Emerging evidence illustrates the imminent role of T cells in sex specific differences in Ang II mediated hypertension. Different types of T cells (pro-hypertensive and antihypertensive) have been found to exhibit gender specific expression. First report came from animal models by Guzik et al. in 2007 who used the male Rag-1−/− mouse model, which lacked mature B and T cells and have a decreased hypertensive response to chronic Ang II infusion and deoxycorticosterone acetate-salt compared to wild type mice.[35] Adoptive transfer of Pan T cells (CD2+CD3+), CD4+, and CD8+ from male donor to male recipient restored the hypertensive response whereas it remained blunted in case of female donor to male recipient or vice versa.[36-38] These findings substantiate the prohypertensive role of specific T cells (CD2+CD3+, CD4+, and CD8) in male mice as compared to females.
In addition, there are antihypertensive T cells called as T regulatory cells (Tregs), previously known as T suppressor cells. Renal T cell profile in females has been found to have greater mRNA expression of the anti-inflammatory Tregs (marker FoxP3) and cytokine IL-10 whereas males have greater population of pro-inflammatory Th17+ CD2+, and CD3+ cells, which can account for the gender differences in hypertension.
In short, the above findings provide some basic insights into the contrasting pathophysiological mechanisms of hypertension in both the sexes. It also calls for more research on immune cells functions, especially to explore their therapeutic potential as antihypertensive agents.
GENDER DISPARITIES IN BP AWARENESS AND CONTROL
A longitudinal study during the time period from 2003–2004 to 2011–2012 reported rising awareness of hypertension in both men and women, with the greatest increase in awareness noted in women.[39] However, at the same time, control of BP in women did not match the rising shift in awareness. Overall women are found to have less controlled BP than men in a cross-sectional analysis of >12,000 patients in primary care visits (54.0% vs. 58.7% P < 0.02), more so in the age group 65–80 even after adjustment for multiple covariates. The Framingham Heart Study also reported more striking age-related decline in hypertension control in women than in men. Possible reasons for poor control of BP in women could be inadequate intensification of BP treatment due to underestimation of CV risk in females by primary care providers; treatment non-adherence; societal discrimination in health-care access; or true treatment resistance.
HYPERTENSION TREATMENT TARGETS – IS THERE A NEED OF GENDER SPECIFIC RECOMMENDATIONS?
European society of hypertension and cardiology guidelines (2018) and AHA guidelines (2017)[40,41] recommend the same BP targets in both men and women despite the plentitude of data reporting sex and gender differences in the pathophysiology and control of hypertension. The recommendation came from the scientific grounds of various trials that found no significant gender differences in CV outcomes. A large meta-analysis on antihypertensive agents using similar cutoffs for men and women showed no gender differences in CV outcomes.[42]
However, whether women are represented in adequate, statistical proportions in majority of trials to confirm the current recommendations are still controversial. A large systematic review of 740 CV clinical trials (2010 and 2017) showed that women cohort represented only 38.2% of trial participants despite women constituting more than 50% of total population.[43,44] In addition, the gender specific data were not generated in most of initial publications as they were primarily not designed or powered to analyze the same. Another confounding factor is majority of these trials directly assessing the impact of BP on CV outcomes included women in menopausal and early postmenopausal age groups (average age 53 years), which limits the generalizability of results to all age groups. As this is not the actual age of the highest prevalence of hypertension among women, the women included in most of the study population were not truly at high risk for CV morbidity and mortality. The recent findings of multi-centric systolic BP intervention trial (SPRINT) reported that more intensive BP control resulted in 25% reduction in CV events.[45] However, women enrollment in SPRINT was only 36% so it failed to draw any statistically significant conclusion on CV reduction with intensification of treatment in women. Therefore, it would not be justifiable to draw any definite gender specific inferences on BP targets and impact of BP control on CV health in women, unless a predetermined analysis is being done to eliminate these confounding variables.
SPECIFIC PHARMACOLOGICAL RECOMMENDATIONS FOR HYPERTENSION IN WOMEN
Before going into details of gender dissimilarities in pharmacotherapy of hypertension, it is important to acknowledge some important sex specific differences in pharmacokinetics and pharmacodynamics of drugs in general.
Women have a higher gastric pH, slower gastric emptying, and a longer total gastrointestinal transit time, compared to men, which can affect the drug bioavailability and absorption. Furthermore, women have lesser weight but higher percentage of body fat than men which can affect the distribution of lipophilic and hydrophilic drugs. Drug distribution in women also gets affected due to their lower plasma volume and subsequently lower average organ blood flow,[46-48] in addition to hormonal influence on drug binding proteins. Hepatic drug metabolism has also cytochrome P450 enzyme variability in both the genders. Women also have 10–25% lower GFR than men even after adjusting for body size which can affect the drug excretion.
The majority of randomized controlled trials on hypertension with CV outcomes have made tremendous strides and provided substantial evidence that BP-lowering treatment is beneficial in both women and men. No strong evidence supported that women and men drive different outcome benefits from specific magnitude of BP reduction or with particular class of antihypertensive drug. Important year-wise landmark trials on hypertension with gender specific CV outcomes are summarized in [Table 1], majority of which show no significant differences in outcomes.[49-54]
| Trial (year) | Study population (age in years) | Total number of patients (women %) | Treatment arms | Results | Gender specific outcomes | |
|---|---|---|---|---|---|---|
| LIFE (2002)[49] | Adults with hypertension and LVH (55–89) | 9193 (54) | Losartan versus atenolol | Losartan superior to atenolol for CV and stroke outcome | Not tested for CV outcome Stroke outcome: No difference |
|
| ALL-HAT (2003)[50] | Adults with hypertension and at least one risk factor for CVD (>55) | 33,357 (47) | Chlorthalidone versus amlodipine/lisinopril | No difference in CV outcome between treatment groups | No sex difference in CV outcome Stroke rate was higher with ACEI than chlorthalidone and amlodipine in women |
|
| VALUE (2006)[51] | Adults at high risk hypertension (>55) | 15,245 (43) | Valsartan versus amlodipine | No difference in CV outcome between treatment groups | CV outcome was superior with amlodipine in women HF outcome was superior with valsartan in men |
|
| ACCOMPLISH (2008)[52] | Adults with systolic hypertension | 11,506 (40) | Hydrochlorothiazide plus benazepril versus amlodipine plus benazepril | ACEI plus CCB is superior than ACEI plus hydrochlorothiazide for CV outcome | No significant gender difference in CV outcomes | |
| HUVET (2008)[53] | Elderly adults (>80) | 3645 (64) | Indapamide with or without ACEI | Indapamide with or without ACEI is associated with reduction in stroke outcome | Data not available | |
| SPRINT (2015)[45] | Adults with hypertension and at increased CV risk (>50) | 9361 (36) | Intensive (SBP<130) versus standard BP target (SBP<140) | Intensive treatment is superior to standard treatment arm for composite CV outcomes | For women subgroup: Hazard ratio was not significant | |
| HYGIA (2019)[54] | Adults with hypertension (>18) | 19,084 (80) | Bedtime drug treatment versus awakening | Bedtime drug treatment is superior to awake drug administration | No significant sex difference | |
LVH: Left ventricular hypertrophy, CV: Cardiovascular, CVD: CV disease, ACE: Angiotensin converting enzyme, ACEI: ACE inhibitor, CCB: Calcium channel blocker, BP: Blood pressure, SBP: Systolic BP
However, the different groups of anti-hypertensive agents exhibit some important pharmacogenomics differences in both the genders [Table 2] that may influence the choice of one specific class of antihypertensive in women than men.[55-66] Similarly, other factors that may affect the selection of one agent over the other are presence of comorbidities such as heart failure and chronic kidney disease and hypertension in reproductive age group where ACEI and ARBs are avoided. Furthermore, women experience more adverse effects with particular antihypertensive medications than men, which may have its own influence in the choice of drugs and treatment adherence.
| Antihypertensive drug class | Sex differences |
|---|---|
| ACEIs[55-57] |
|
| ARBs[56-58] |
|
| CCBs[59-61] |
|
| Beta blockers[62-64] |
|
| Thiazide diuretics[65,66] |
|
ARBs: Angiotensin receptor blockers, CCBs: Calcium channel blockers, ACE: Angiotensin converting enzyme, ACEIs: ACE inhibitors, BP: Blood pressure
Some important practical points to consider are as follows:
Diuretic therapy may be more beneficial in postmenopausal hypertensive women as it reduces urinary calcium excretion and prevents bone loss.
Calcium channel blockers (CCBs) have been found to be more effective than ACE inhibitors for stroke prevention in women. Hence, CCBs can be the preferred agents in women at higher stroke risk.
Avoid thiazides in women at risk of electrolyte imbalance.
CCBs-induced edema and ACEI-induced cough are more marked in women than men and can be taken into consideration in corresponding clinical scenarios.
To summarize, though no significant gender treatment interaction has been noticed as far as CV outcomes or drug recommendations are concerned, minor noteworthy differences found in various studies deserve further analysis to draw valid conclusions, especially when existing literature propose gender related incongruities in the various other clinical aspects of hypertension.
Table 3 outlines the key points in relation to hypertension in women discussed so far.
| Female sex/gender specific distinction | |
|---|---|
| Epidemiology |
|
| Pathophysiology |
|
| CV impact of hypertension |
|
| Awareness and control |
|
| BP treatment targets |
|
| Anti-hypertensive drug effects |
|
| Future goals |
|
RAS: Renin angiotensin system, CCBs: Calcium channel blockers, BP: Blood pressure, CV: Cardiovascular
CONCLUSION
In this progressive era of personalized treatment where we are focusing on every other associate of hypertension such as age, ethnicity, and diabetes, gender/sex specific attributes of hypertension have not yet been precisely addressed on scientific grounds. Hypertension in women is a dynamic process and a stimulating area of preventive CV medicine, which needs further in depth research on priority basis so as to curb this global burden of death and disability in women.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupation-al, and metabolic risks or clusters of risks for 195 countries and territories 1990-2017: A systematic analysis for the global burden of disease study 2017. Lancet. 2018;392:1923-94.
- [CrossRef] [Google Scholar]
- Risk factors for sudden cardiac death in post-menopausal women. J Am Coll Cardiol. 2012;60:2674-82.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular mortality associated with 5 leading risk factors: national and state preventable fractions estimated from survey data. Ann Intern Med. 2015;163:245-53.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence, awareness, and management of hypertension, dyslipidemia, and diabetes among United States adults aged 65 and older. J Gerontol A Biol Sci Med Sci. 2009;64:256-63.
- [CrossRef] [PubMed] [Google Scholar]
- Report of the Public Health Service Task Force on Women's Health Issues. Public Health Rep. 1985;100:73-106.
- [Google Scholar]
- Fact Sheet. 2019. Geneva: World Health Organization; Available from: https://www.who.int/news-room/fact-sheets/detail/hypertension [Last accessed on 2020 Jan 25]
- [Google Scholar]
- Socioeconomic and demographic predictors of high blood pressure, diabetes, asthma and heart disease among adults engaged in various occupations: Evidence from India. J Biosoc Sci. 2020;52:629-49.
- [CrossRef] [PubMed] [Google Scholar]
- Report on Medical Certification of Cause of Death. 2017. New Delhi: Registrar General of India; Available from: https://www.censusindia.gov.in/2011-documents/mccd_report1/MCCD_report-2017.pdf [Last accessed on 2020 Jun 27]
- [Google Scholar]
- Gender differences in the regulation of blood pressure. Hypertension. 2001;37:1199-208.
- [CrossRef] [PubMed] [Google Scholar]
- Sex differences in hypertension: Where we have been and where we are going. Am J Hypertens. 2018;31:1247-54.
- [CrossRef] [PubMed] [Google Scholar]
- Prehypertension: A meta-analysis of the epidemiology, risk factors, and predictors of progression. Tex Heart Inst J. 2011;38:643-52.
- [Google Scholar]
- Sex differences in prevalence and risk factors of hypertension in India: Evidence from the national family health survey. PLoS One. 2021;16:e0247956.
- [CrossRef] [PubMed] [Google Scholar]
- 2017 ACC/AHA guidelines for BP management in adult patients. Pharm Today. 2018;24:57-73.
- [CrossRef] [Google Scholar]
- Role of ambulatory blood pressure monitoring in predicting adverse maternofetal events in pregnancy. Indian J Cardiovasc Dis Women WINCARS. 2021;6:17-24.
- [CrossRef] [Google Scholar]
- Global disparities of hypertension prevalence and control: A systematic analysis of population-based studies from 90 countries. Circulation. 2016;134:441-50.
- [CrossRef] [PubMed] [Google Scholar]
- Sex-specific differences in cardiovascular risk factors and blood pressure control in hypertensive patients. J Clin Hypertens (Greenwich). 2014;16:309-12.
- [CrossRef] [PubMed] [Google Scholar]
- Assessing sex differences in the risk of cardiovascular disease and mortality per increment in systolic blood pressure: A systematic review and meta-analysis of follow-up studies in the United States. PLoS One. 2017;12:e0170218.
- [CrossRef] [PubMed] [Google Scholar]
- Increased blood pressure variability in menopause. Eur Rev Med Pharmacol Sci. 2008;12:89-95.
- [Google Scholar]
- Stress, menopausal status and nocturnal blood pressure dipping patterns among hypertensive women. Can J Cardiol. 2009;25:e157-63.
- [CrossRef] [Google Scholar]
- Optimal indicators of home BP variability in perimenopausal women and associations with albuminuria and reproducibility: The J-HOT home BP study. Am J Hypertens. 2015;28:586-94.
- [CrossRef] [PubMed] [Google Scholar]
- Differences between men and women in ambulatory blood pressure thresholds for diagnosis of hypertension based on cardiovascular outcomes. Chronobiol Int. 2013;30:221-32.
- [CrossRef] [Google Scholar]
- Heart disease and stroke statistics-2016 update: A report from the American heart association. Circulation. 2016;133:e38-360.
- [Google Scholar]
- Sex differences in hypertension and stroke risk in the REGARDS study: A longitudinal cohort study. Hypertension. 2019;74:749-55.
- [CrossRef] [PubMed] [Google Scholar]
- Gender-specific association between the blood pressure category according to the updated ACC/AHA guidelines for hypertension and cardio-ankle vascular index: A community-based cohort study. J Cardiol. 2020;75:578-82.
- [CrossRef] [PubMed] [Google Scholar]
- Arterial stiffness as a measure of vascular dysfunction in hypertensive women. Ind J Cardiovasc Dis Women. 2020;5:308-12.
- [CrossRef] [Google Scholar]
- Thresholds for conventional and home blood pressure by sex and age in 5018 participants from 5 populations. Hypertension. 2014;64:695-701.
- [CrossRef] [PubMed] [Google Scholar]
- The protective role of estrogen and estrogen receptors in cardiovascular disease and the controversial use of estrogen therapy. Biol Sex Differ. 2017;8:33.
- [CrossRef] [PubMed] [Google Scholar]
- Sex, the brain and hypertension: Brain oestrogen receptors and high blood pressure risk factors. Clin Sci (Lond). 2016;130:9-18.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular risk in women: Focus on hypertension. Can J Cardiol. 2014;30:553-9.
- [CrossRef] [PubMed] [Google Scholar]
- Hypertension in premenopausal women: Is there any difference? In: High Blood Press Cardiovasc Prev. Vol 21. 2014. p. :195-9.
- [CrossRef] [PubMed] [Google Scholar]
- Structure and functions of angiotensinogen. Hypertens Res. 2016;39:492-500.
- [CrossRef] [PubMed] [Google Scholar]
- Is the renin-angiotensin system actually hypertensive? Pediatr Nephrol. 2014;29:951-60.
- [CrossRef] [PubMed] [Google Scholar]
- A lower ratio of AT1/AT2 receptors of angiotensin II is found in female than in male spontaneously hypertensive rats. Cardiovasc Res. 2004;62:587-93.
- [CrossRef] [PubMed] [Google Scholar]
- Angiotensin (1-7) receptor antagonism equalizes angiotensin II-induced hypertension in male and female spontaneously hypertensive rats. Hypertension. 2010;56:658-66.
- [CrossRef] [PubMed] [Google Scholar]
- Role of the t cell in the genesis of angiotensin ii induced hypertension and vascular dysfunction. J Exp Med. 2007;204:2449-60.
- [CrossRef] [PubMed] [Google Scholar]
- Sex-specific T-cell regulation of angiotensin II-dependent hypertension. Hypertension. 2014;64:573-82.
- [CrossRef] [PubMed] [Google Scholar]
- Sex differences in T-lymphocyte tissue infiltration and development of angiotensin II hypertension. Hypertension. 2014;64:384-90.
- [CrossRef] [PubMed] [Google Scholar]
- Sex-specific immune modulation of primary hypertension. Cell Immunol. 2015;294:95-101.
- [CrossRef] [PubMed] [Google Scholar]
- Trends in blood pressure among adults with hypertension: United States, 2003 to 2012. Hypertension. 2015;65:54-61.
- [CrossRef] [PubMed] [Google Scholar]
- 2017 ACC/AHA/AAPA/ABC/ACPM/ AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: A report of the American college of cardiology/American heart association task force on clinical practice guidelines. J Am Coll Cardiol. 2018;71:127-248.
- [CrossRef] [PubMed] [Google Scholar]
- ESC/ESH Guidelines for the management of arterial hypertension. Eur Heart J. 2018;39:3021-104.
- [CrossRef] [PubMed] [Google Scholar]
- Do men and women respond differently to blood pressure-lowering treatment? Results of prospectively designed overviews of randomized trials. Eur Heart J. 2008;29:2669-80.
- [CrossRef] [PubMed] [Google Scholar]
- Women's participation in cardiovascular clinical trials from 2010 to 2017. Circulation. 2020;141:540-8.
- [CrossRef] [PubMed] [Google Scholar]
- US Census Bureau Quick Facts. 2019. Available from: https://www.census.gov/quickfacts/fact/table/US/PST045219 [Last accessed on 2021 Jan 28]
- [Google Scholar]
- A randomized trial of internsive versus standard blood-pressure control. N Engl J Med. 2015;373:2103-16.
- [CrossRef] [PubMed] [Google Scholar]
- Pharmacological differences between men and women In: Huang SM, Lertora A, Atkinson J, Markey S Jr, eds. Principles of Clinical Pharmacology (3rd ed). Cambridge, Massachusetts: Academic Press; 2012. p. :383-94.
- [CrossRef] [PubMed] [Google Scholar]
- Sex differences in drug effects: Interaction with sex hormones in adult life. Handb Exp Pharmacol. 2012;214:91-105.
- [CrossRef] [PubMed] [Google Scholar]
- Does sex matter? The influence of gender on gastrointestinal physiology and drug delivery. Int J Pharm. 2011;415:15-28.
- [CrossRef] [PubMed] [Google Scholar]
- Stroke reduction in hypertensive adults with cardiac hypertrophy randomized to losartan versus atenolol: The losartan intervention for endpoint reduction in hypertension study (LIFE) Hypertension. 2005;45:46-52.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical events in high-risk hypertensive patients randomly assigned to calcium channel blocker versus angiotensin-converting enzyme inhibitor in the antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALL-HAT) Hypertension. 2006;48:374-84.
- [CrossRef] [PubMed] [Google Scholar]
- Outcomes in subgroups of hypertensive patients treated with regimens based on valsartan and amlodipine: An analysis of findings from the VALUE trial. J Hypertens. 2006;24:2163-8.
- [CrossRef] [PubMed] [Google Scholar]
- Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high-risk patients (ACCOMPLISH) N Engl J Med. 2008;359:2417-28.
- [CrossRef] [PubMed] [Google Scholar]
- Treatment of hypertension in patients 80 years of age or older (HUVET) N Engl J Med. 2008;358:1887-98.
- [CrossRef] [PubMed] [Google Scholar]
- Bedtime hypertension treatment improves cardiovascular risk reduction: The Hygia chronotherapy trial. Eur Heart J. 2020;41:4565-76.
- [CrossRef] [PubMed] [Google Scholar]
- Gender differences in angiotensin-converting enzyme (ACE) activity and inhibition by enalaprilat in healthy volunteers. J Cardiovasc Pharmacol. 2004;43:737-44.
- [CrossRef] [PubMed] [Google Scholar]
- Reporting on sex-based analysis in clinical trials of angiotensin-converting enzyme inhibitor and angiotensin receptor blocker efficacy. Can J Cardiol. 2008;24:491-6.
- [CrossRef] [Google Scholar]
- Antihypertensive drug therapy and blood pressure control in men and women: An international perspective. J Hum Hypertens. 2010;24:336-44.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of the relationship between sex, polymorphisms in CYP2C8 and CYP2C9, and pharmacokinetics of angiotensin receptor blockers. Drug Metab Dispos. 2013;41:224-9.
- [CrossRef] [PubMed] [Google Scholar]
- Sexand age-related antihypertensive effects of amlodipine. The amlodipine cardiovascular community trial study group. Am J Cardiol. 1996;77:713-22.
- [CrossRef] [Google Scholar]
- Gender-specific effects on verapamil pharmacokinetics and pharmacodynamics in humans. J Clin Pharmacol. 2000;40:219-30.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of gender on the pharmacokinetics of verapamil and norverapamil in human. Biopharm Drug Dispos. 2006;27:329-34.
- [CrossRef] [PubMed] [Google Scholar]
- Gender-related effects on metoprolol pharmacokinetics and pharmacodynamics in healthy volunteers. Clin Pharmacol Ther. 1999;66:594-601.
- [CrossRef] [Google Scholar]
- Metoprolol dose equivalence in adult men and women based on gender differences: Pharmacokinetic modeling and simulations. Med Sci (Basel). 2016;4:18.
- [CrossRef] [PubMed] [Google Scholar]
- Tolerability of beta-blockers metabolized via cytochrome P450 2D6 is sex-dependent. Clin Pharmacol Ther. 2006;80:551-3.
- [CrossRef] [PubMed] [Google Scholar]
- Blood pressure-lowering efficacy of monotherapy with thiazide diuretics for primary hypertension. Cochrane Database Syst Rev. 2014;29:CD003824.
- [CrossRef] [PubMed] [Google Scholar]
- Thiazide-associated hyponatremia: A population-based study. Am J Kidney Dis. 2013;62:67-72.
- [CrossRef] [PubMed] [Google Scholar]







