Translate this page into:
Unilateral Transudative Pleural Effusion due to rare Cause – A Case Report
-
Received: ,
Accepted: ,
How to cite this article: Bhaskar K, Devanandan K, Jain JK, Krishna T, Gongati P. Unilateral transudative pleural effusion due to rare cause – A case report. Indian J Cardiovasc Dis Women 2022;7:149-52.
Abstract
Transudative pleural effusions are characterized by low protein and lactate dehydrogenase according to Light’s criteria. Common causes are congestive cardiac failure, nephrotic syndrome, liver cirrhosis, and protein-losing enteropathy. Constrictive pericarditis is a rare cause of transudative pleural effusion. It can cause bilateral or unilateral recurrent effusions and chylothorax rarely. Common causes of constrictive pericarditis include infections such as viruses and tuberculosis, radiation, and cardiac surgery. We present a treated case of pulmonary tuberculosis presenting with massive, left-sided, and transudative pleural effusion due to constrictive pericarditis diagnosed by computed tomography scan and 2D echo with classical findings. Incidental findings were internal jugular vein thrombus and right lower lobar pulmonary embolus. This case highlights the atypical presentation of constrictive pericarditis as unilateral pleural effusion. A proper history with a high index of suspicion is essential in the workup of constrictive pericarditis which can be cured by pericardiectomy.
Keywords
Constrictive pericarditis
Tuberculosis
Pulmonary embolism
INTRODUCTION
Pleural effusions can be exudative and transudative. Exudative effusions are characterized by elevated protein and lactate dehydrogenase (LDH) as defined by Light’s criteria and are commonly caused by infectious, inflammatory conditions, and malignancies. Transudative pleural effusions are formed by changes in starling’s forces, that is, hydrostatic and oncotic pressures of the intravascular compartment and are characterized by low protein, cells, and LDH. Common causes of transudative pleural effusions include congestive heart failure, renal, hepatic failure, and hypoproteinemia. Rare causes include peritoneal dialysis, nephrotic syndrome, Meigs syndrome, and urinothorax. Hereby, we present a case of constrictive pericarditis due to post-tuberculous sequelae, leading to the left unilateral massive transudative pleural effusion.
CASE REPORT
A 43-year-old male patient came to the outpatient department with the complaints of breathlessness and cough with minimal mucoid sputum of 3 months. He had lost appetite and weight of approximately 10 kg in the preceding 1½ months. There was no orthopnea, paroxysmal nocturnal dyspnea, pedal edema, or abdominal distension. He had a history of pulmonary tuberculosis 1 year back for which he was given antitubercular therapy for 6 months with symptomatic improvement. He was hypertensive on regular medication. He was not a diabetic. He was a known case of hyperthyroidism receiving carbimazole. He was a non-smoker and social alcoholic. At the time of presentation, he was conscious and coherent, his saturation was 98% with room air, respiratory rate was 18 per min, and blood pressure 106/60 mmHg. On examination, JVP was elevated with prominent rapidly collapsing y descent, Kussmaul’s sign was positive. On auscultation, S2 was wide split and the pericardial knock was heard. Lung auscultation revealed left-sided diminished breath sounds. His sputum was negative for acid-fast bacilli, Gram staining, and culture. Hemoglobin was 11.3 g/dL, TC 8400 cells/cu mm, ESR 10 mm/1st h, and D-dimer 771 µg/m/mL. His liver and renal function tests were normal. Viral markers were negative, thyroid: T3 – 0.8 n mol/L (1.1–3.1), T4 – 5.16 mg/dL (4.5–11.7), and TSH – 9.32 m IU/mL (0.2–4), connective tissue profile was negative, his chest x-ray showed left-sided pleural effusion. His pleural fluid analysis showed glucose 115 mg/dL, protein 2.7 g/dL (serum 5.6 g/dl), albumin 1.4 g/d L (serum 2.8 g/dl), LDH 167 U/L (pleural fluid serum ratio: 0.52), amylase 43U/L, RBC: 1920 cells, differential count: 320 cells/Cu mm with polymorphs 70%, lymphocytes 30%, and ADA 11 IU/L suggestive of transudative pleural effusion. CECT thorax showed internal jugular, superior vena cava thrombi, and right lower lobe pulmonary artery embolism [Figures 1 and 2]. Bilateral lower limb venous Doppler was normal. A 2D echo showed tachycardia, Grade 2 left ventricular diastolic dysfunction, and paradoxical interventricular septum, significant respiratory variation is seen across mitral and tricuspid valves, inferior vena cava dilated and non-collapsible, thickened pericardium of 5 mm suggestive of chronic constrictive pericarditis [Figure 3]. FDG PET scan showed mediastinal adenopathy with SUV max 2.8 and FDG avid pericardial thickening [Figure 4]. The patient was advised for an endobronchial ultrasound-guided mediastinal lymph node biopsy for further workup. He was advised for pericardiectomy. The current medications include antituberculous therapy and anticoagulation for pulmonary thromboembolism. An endocrinologist consultation was taken for hyperthyroidism. The patient is under follow-up with a stable clinical condition.
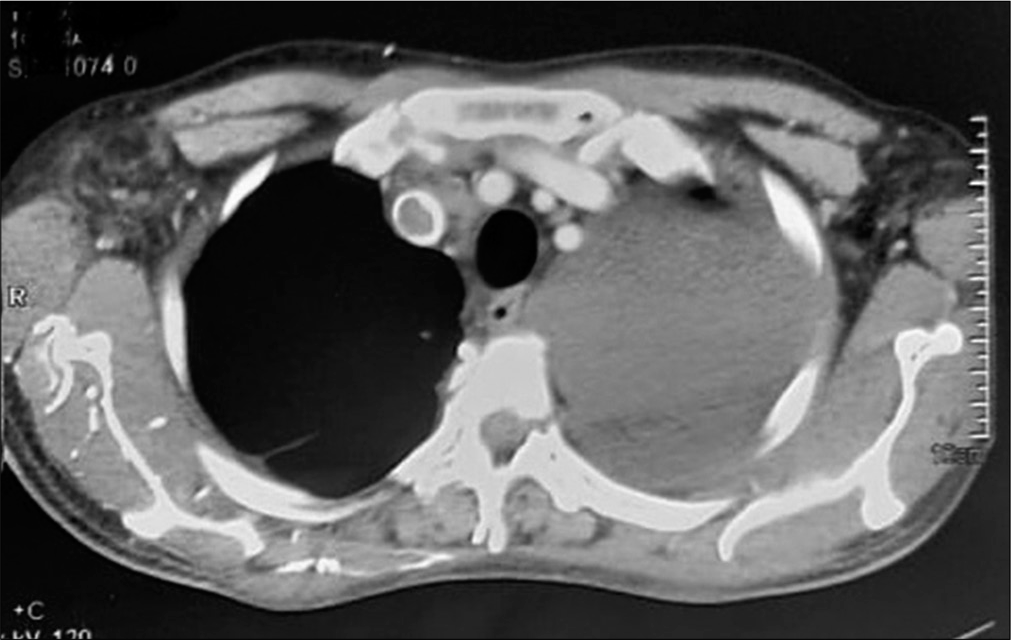
- CECT thorax shows superior vena cava thrombosis and left massive pleural effusion.
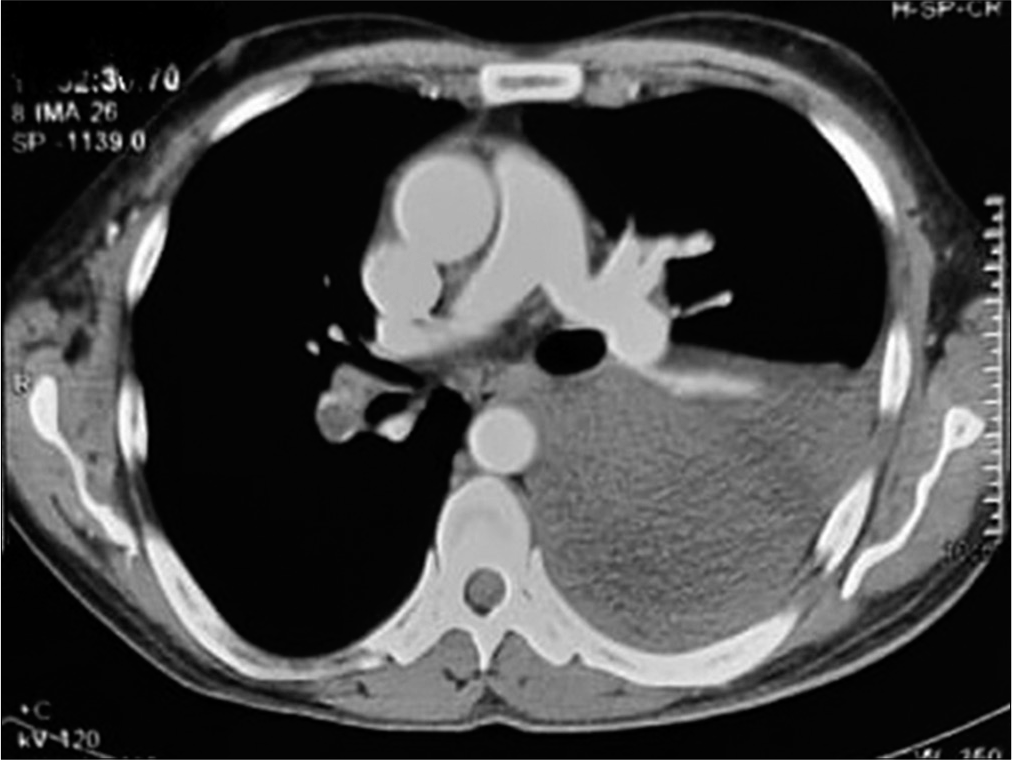
- Right lower lobar pulmonary artery thrombus.
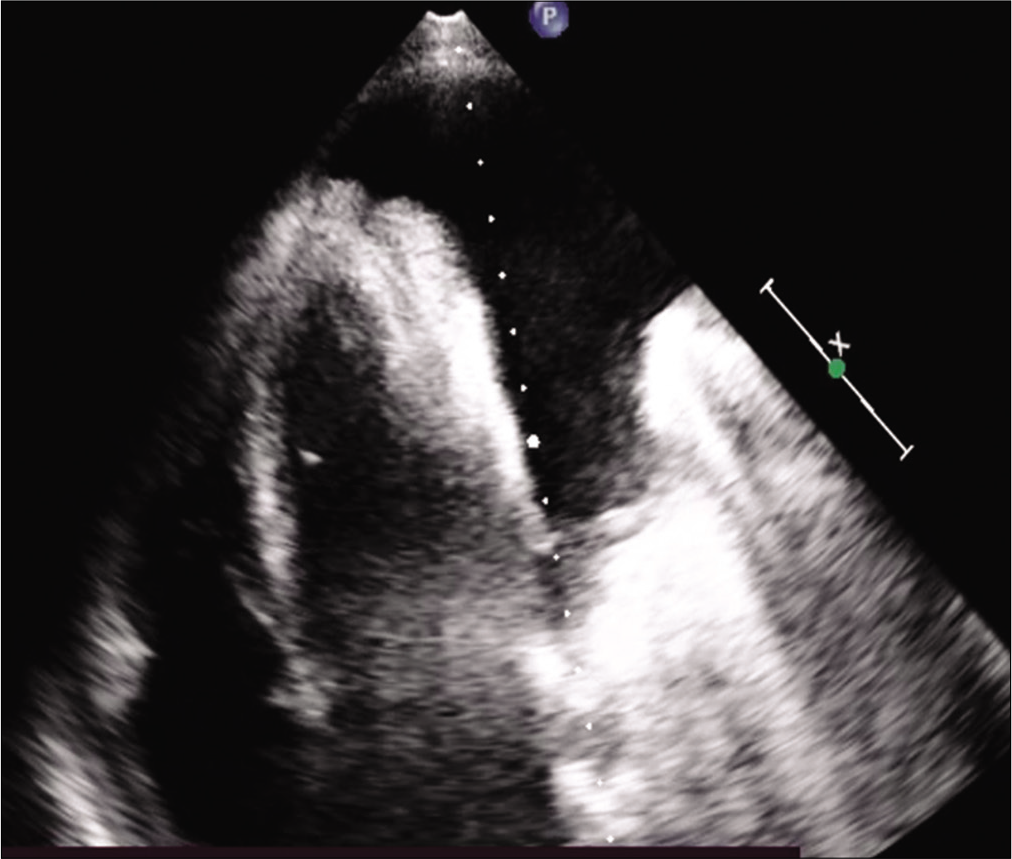
- Apical 4-chamber view.
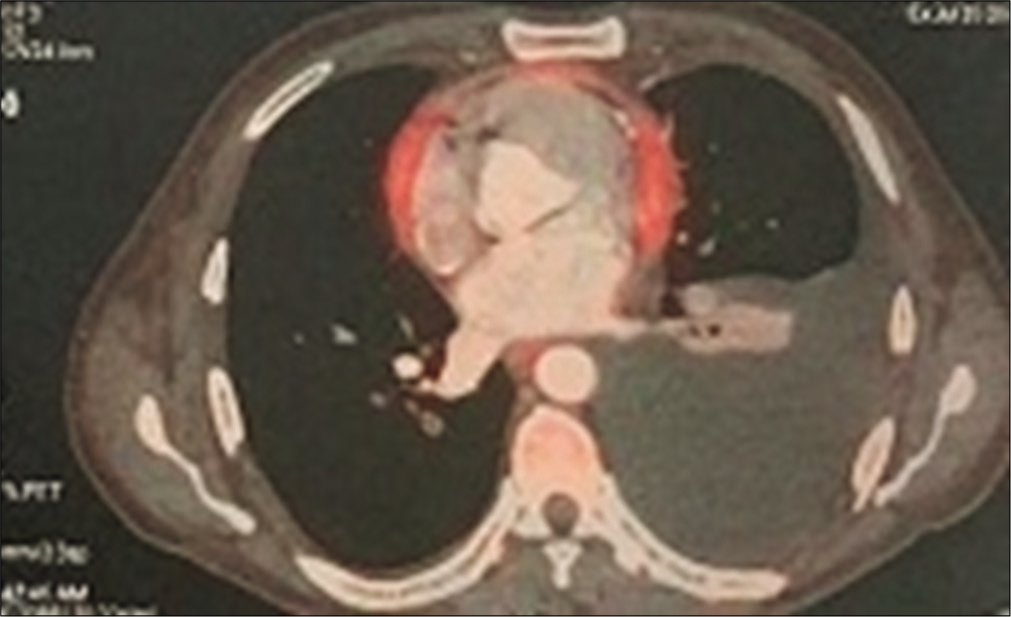
- PET computed tomography showing thick, active the pericardium.
CASE DISCUSSION
Common causes of transudative pleural effusion include congestive heart failure, cirrhosis of the liver, hypoproteinemia, and nephrotic syndrome. Rare causes include urinothorax, myxedema, and cerebrospinal fluid leaks. Light’s criteria are used to differentiate exudative pleural effusions from transudates. The presence of one of the three criteria: Pleural fluid and serum protein ratio of more than 0.5, pleural fluid and serum LDH ratio of more than 0.6, and pleural fluid LDH of more than 2/3 of the upper limit of the laboratory’s reference range of serum LDH suggest exudative effusion.[1] CHF causes bilateral or right-sided transudative effusions typically. A 60% incidence of pleural effusion has been reported in constrictive pericarditis cases, 2/3 bilateral, and symmetrical. Rarely, constrictive pericarditis can present as a unilateral recurrent pleural effusion.[2-4] Constrictive pericarditis leads to bilateral[5] mild effusions commonly, moderate to massive effusions are rare.[6] Biochemically, these are commonly exudates but transudative effusions are also reported.[2] Chylothorax can also present as an unusual sequela of constrictive pericarditis.[7]
Constrictive pericarditis is characterized by thickened pericardium without associated effusion. Effusive constrictive pericardial effusion is characterized by concurrent pericardial effusion and constricted pericardium.[8] A common presentation of CP is congestive heart failure.[9] Common causes of constrictive pericarditis include tuberculous (46.8%),[10,11] post-cardiac surgery,[12] radiation,[10,12] uremia,[10] post-traumatic,[10] and idiopathic.[10,12] Etiology can vary depending on the center, as post-surgical and radiation dominate in referral centers.[9] Impaired diastolic filling of ventricles is the main pathophysiological feature.[9,13] This leads to increased hydrostatic pressure resulting in transudative pleural effusions. Pericardial thickening is the classical finding and diagnostic although in 20% of cases, thickening was not a prominent finding.[13]
Our patient had a history of pulmonary tuberculosis and pulmonary fibrosis on imaging. Constrictive pericarditis supported by classical 2D echo and computed tomography (CT) findings of pericardial thickening strongly favors tuberculosis etiology for all these manifestations.
Pulmonary embolism is also a possible cause of transudative effusion in our patients. In a study, 47% of patients with pulmonary embolism had pleural effusions in CT scans.[14] However, the presence of effusion was not an independent risk factor for mortality in pulmonary embolism.[15] These effusions were exudative in almost all cases.[14,16] Infarction was not related to the pleural effusion.[14] However, in our case, the presence of pulmonary embolism on the right side makes it an unlikely cause of the left-sided pleural effusion.
The reason for the extensive jugular vein thrombus and pulmonary embolism is again attributed due to ongoing active inflammation as evidenced by active pericardial uptake in PET scan. Tuberculosis is a hypercoagulable state as evidenced by several studies in the literature.[17-19] Other causes of hypercoagulable states like malignancy have been ruled out by PET scan and negative connective tissue profile.
Although FDG uptake in pericardium indicates ongoing inflammation, transudative instead of exudative pleural effusion suggests that the mechanism of pleural fluid formation is the result of changes in the hydrostatic pressure rather than inflammatory.
Pericardiectomy is the definitive treatment modality. It is associated with high mortality and morbidity.[10] Tuberculosis as an etiology has no impact on mortality.[11] Advanced age, atrial fibrillation, concomitant tricuspid insufficiency, inotropic support, and low cardiac output were significant negative predictors of survival.[20]
The simultaneous presence of pulmonary embolism and constrictive pericarditis in our patient makes this case unique. Drawbacks in our case report are as follows: No histological proof of tuberculosis at this presentation, cardiac catheterization, and hemodynamic studies could not be conducted.
CONCLUSION
Constrictive pericarditis should be considered in the differential diagnosis of unilateral transudative pleural effusion and a history of the previous infections may provide a diagnostic clue. While bilateral pleural effusions, chronic pleural effusions, and recurrent pleural effusions are common presentations of constrictive pericarditis in the literature, this left-sided, massive, and transudative effusion in association with pulmonary thromboembolism made our case unique.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- The undiagnosed pleural effusion. Clin Chest Med. 2006;27:309-19.
- [CrossRef] [PubMed] [Google Scholar]
- Constrictive pericarditis presenting as pleural effusion of unknown origin. Arch Intern Med. 1989;149:201-3.
- [CrossRef] [PubMed] [Google Scholar]
- A 47-year-old man with recurrent unilateral pleural effusion. Chest. 2017;152:e121-4.
- [CrossRef] [PubMed] [Google Scholar]
- A 43-year-old man with a large recurrent right-sided pleural effusion. Chest. 2000;117:1191-4.
- [CrossRef] [PubMed] [Google Scholar]
- Constrictive pericarditis presenting as bilateral pleural effusion: A report of two cases. Cureus. 2018;10:e2451.
- [CrossRef] [PubMed] [Google Scholar]
- Massive right pleural effusion and ascites caused by a primary constrictive pericardial band. Jpn J Thorac Cardiovasc Surg. 2002;50:350-2.
- [CrossRef] [PubMed] [Google Scholar]
- Constrictive pericarditis is a rare cause of chylothorax: A case report. Eur Heart J Case Rep. 2018;2:yty113.
- [CrossRef] [PubMed] [Google Scholar]
- Effusive-constrictive pericarditis: Current perspectives. J Vasc Diagn Interv. 2018;6:7-14.
- [CrossRef] [Google Scholar]
- Constrictive pericarditis in the Modern Era. Circulation. 1999;100:1380-6.
- [CrossRef] [PubMed] [Google Scholar]
- Long-term outcomes of pericardiectomy for constrictive pericarditis. J Cardiothorac Surg. 2015;10:177.
- [CrossRef] [PubMed] [Google Scholar]
- Constrictive pericarditis: 21 years' experience and review of the literature. Pan Afr Med J. 2021;38:141.
- [CrossRef] [PubMed] [Google Scholar]
- Constrictive pericarditis: Aetiology and cause-specific survival after pericardiectomy. J Am Coll Cardiol. 2004;43:1445-52.
- [CrossRef] [PubMed] [Google Scholar]
- 2015 ESC guidelines for the diagnosis and management of pericardial diseases. Eur Heart J. 2015;36:2921-64.
- [CrossRef] [PubMed] [Google Scholar]
- Analysis of pleural effusions in acute pulmonary embolism: Radiological and pleural fluid data from 230 patients. Respirology. 2007;12:234-9.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence and clinical significance of pleural effusion in patients with acute pulmonary embolism: A retrospective study. J Thorac Dis. 2021;13:541-51.
- [CrossRef] [PubMed] [Google Scholar]
- Pleural effusions in pulmonary emboli: A single centre experience. Cureus. 2020;12:11942.
- [CrossRef] [PubMed] [Google Scholar]
- Pulmonary tuberculosis induces a systemic hypercoagulable state. J Infect. 2015;70:324-34.
- [CrossRef] [PubMed] [Google Scholar]
- Acute-phase response and the hypercoagulable state in pulmonary tuberculosis. Br J Haematol. 1996;93:943-9.
- [CrossRef] [PubMed] [Google Scholar]
- Tuberculosis and venous thromboembolism: A case series. Cases J. 2009;2:1-4.
- [CrossRef] [PubMed] [Google Scholar]
- Pericardiectomy for chronic constrictive tuberculous pericarditis. Tex Heart Inst J. 2003;30:180-5.
- [Google Scholar]







