Translate this page into:
Cardiac MRI in Evaluation of Dilated Cardiomyopathy
Jyotsna Rani, MD, Professor Department of Radiology & Imageology, Nizams Institute of Medical Sciences Hyderabad, Telangana 500082 India yjyotsna@yahoo.com
This article was originally published by Thieme Medical and Scientific Publishers Private Ltd. and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Abstract
Cardiac magnetic resonance imaging (MRI) plays an important role in the evaluation of dilated cardiomyopathy, and is the gold-standard technique in identifying the various etiological factors and differentiating one from the other. It not only helps in identifying the cause but also aids in monitoring the treatment response. The present article reviews the different MRI techniques available, recent advancements, and limitations in technology in the evaluation of patients with dilated cardiomyopathy in the present-day scenario.
Keywords
dilated cardiomyopathy
late gadolinium enhancement
left ventricular dysfunction
Introduction
Dilated cardiomyopathy (DCM) is the cardiac muscle disorder occurs due to genetic, infective, inflammatory, or drug-induced reasons, leading to left-ventricular dilation and systolic dysfunction.1 2 3 4 5 It can have varied clinical manifestations from an asymptomatic state, heart failure to arrhythmias and death.6
Cardiac magnetic resonance imaging (CMRI) is currently considered the gold standard due to high temporal and excellent spatial resolution, enabling accurate assessment. The three-dimensional (3D) dataset available with MRI provides a more precise and reproducible method for quantifying ventricular volumes, mass, and function in patients with DCM. This technique provides insight into ventricular morphology, function, and myocardial tissue characterization. Tissue characterization with delayed contrast enhancement (CE) provides insight into the etiology of several myocardial diseases. It is also used to prognosticate the patient of DCM, as left-ventricular ejection fraction (LVEF) can be calculated accurately.7
Transthoracic two-dimensional (2D) echo (TTE; sometimes transesophageal echo [TEE], when there is a poor echo window) is helpful to know the severity of left-ventricular dysfunction (LVD) and degree of LV dilatation (end-diastolic diameter > 55 mm), and may provide information to determine the cause of DCM.8 However with MRI, we can gather additional information, such as single-photon emission computed tomography myocardial perfusion scintigraphy (SPECT MPS), glucose metabolism on the F-18 fluorodeoxyglucose (18F-FDG) images, and cardiac CT rules out the ischemic component in DCM.
Delayed contrast material, enhanced cardiac magnetic resonance (MR) imaging, is used to detect both viability and myocardial infarction (MI). But its usage is increased in nonischemic cardiomyopathies,9 as the percentage of viable myocardium in this condition is also an important prognostic factor for mortality. In DCM, MRI provides a clue to take the biopsy for the diagnosis of secondary causes, when required.10
MRI in DCM
MRI is useful in DCM in multiple ways. It helps in differentiating ischemic from nonischemic cardiomyopathy and varieties of nonischemic forms, assessing both ventricular functions while determining CRT (cardiac resynchronize therapy), detecting thrombus, and prognosticating DCM.
MR Technique
There are changes in morphology in the form of dilatation of ventricular chambers and functional changes as well in a patient with dilated cardiomyopathy. Therefore, MRI protocols in the evaluation of dilated cardiomyopathy should be tailored to assess the morphology and functional status of the heart to arrive at a diagnosis.11 12
Cardiac MRI uses seven basic pulse sequences for the assessment of cardiovascular disease as mentioned below:
-
Black blood imaging for the assessment of cardiac morphology.
-
Bright blood imaging for creating cine and images of the heart.
-
Fat saturation imaging for the assessment of fatty infiltration of cardiac masses and myocardium.
-
Velocity encoding mapping for the measurement of blood flow.
-
Perfusion imaging with gadolinium for the evaluation of vascularity.
-
Inversion recovery late hyperenhancement (LHE) with gadolinium for the assessment of abnormal myocardial tissue composition 0.1 to 0.2 mmol/kg with a delay of 10 to 15 minutes which nulls the normal myocardial signal, so that pathological changes would be highlighted, and7 angiography with gadolinium for the assessment of the vasculature.13
-
3D whole-heart navigator-gated steady-state free precession (SSFP) for vascular anatomy, including coronary arteries, can be studied without contrast.14
Basic Cardiac MRI
The basic protocols include a four-chamber long axis, two-chamber vertical axis, and short-axis views using breath-hold cine SSFP sequences covering both ventricles.
In DCM, the static images by the spin-echo sequence, represented as black blood, are used to measure the dimensions of the LV. Cine loss by the gradient-echo is useful to quantify the severity of LV dysfunction. T1 or T2 contrast by preparation pulses are used for tissue characterization like edema, fibrosis, or different infiltrative disorders.
Fast and real-time sequences include fast low-angle shot, spiral, and echo-planar imaging.
MRI involves quantifying ventricular volumes, mass, and function in patients with DCM. Black and bright blood images provide information about the morphology of the heart. Gradient-echo sequences with an encoding of velocity are used for velocity and flow. DCM patients usually have heterogeneous end-diastolic LV wall thickness, and there is loss of the physiological gradient in systolic wall thickening between LV basal and apical segments. Fast gradient echo sequence can be used along with the T1-mapping technique to detect diffuse myocardial fibrosis. Fatty infiltration at particular chambers of the heart can be detected with fat saturation imaging to diagnose arrhythmogenic right ventricular dysplasia.
T1-weighted black blood is used for myocardial fat imaging, and T2-weighted short-tau inversion recovery (T2w-STIR) imaging using an ECG-gated triple inversion recovery (IR) technique is utilized to observe changes of tissue edema. The two- and four-chamber views are obtained for assessment of the entire heart. Abnormal myocardium appears bright in contrast to normal myocardium. The abnormal bright region can come due to edema, necrosis, fibrosis, or various infiltrates which expand the extracellular space of the myocardium.13 14 By T2-mapping technique, amount of intramyocardial iron deposition in hemochromatosis can be estimated.
Thrombus is suspected when there is a soft tissue intracavity mass, which does not enhance after contrast. and has variable signal intensity on either T1 or T2 images.
Velocity Encoding Imaging
To decrease the long-scan times for flow by MRI, a combination of phase contrast MRI fast sampling strategies, such as spiral or radial imaging, and other imaging strategies, such as balanced SSFP emerged. But, in velocity encoding, noise reduction along with increased spatial and/or temporal resolution is useful for the assessment of left ventricular performance (e.g., cardiac output) and regurgitation volumes. Secondary MRI in DCM can also be assessed by this method.
MR Perfusion Images with Contrast
Myocardial perfusion can be studied with contrast (Table 1).
|
MRI technique |
Information |
|---|---|
|
Abbreviations: ECV, extracellular volume; IR-CE, inversion recovery contrast-enhanced; LGE, late gadolinium enhancement; MR, magnetic resonance; PVR, perspective volume rendering; SSFP. steady state free precession; T2w-STIR, T2-weighted short-tau inversion recovery. |
|
|
Dynamic first-pass perfusion images |
Perfusion defects or microvascular dysfunction |
|
Delayed perfusion images (IR-CE) |
LGE–scar and fibrosis |
|
T1-mapping techniques with contrast |
Can measure ECV–a biomarker of fibrosis.13 |
|
T2w-STIR imaging |
To detect the tissue edema, which is a marker of active inflammation |
|
Cine SSFP or tagging techniques |
For evaluation of ventricular function and dyssynchrony |
|
PVR of MR |
Imaging datasets to simulate endoscopic views of human organ systems. |
|
Phase-contrast sequences |
For assessment of valvular regurgitation and diastolic function |
|
MR angiography |
Vascular anatomy |
Patterns of Delayed Enhancement
Gadolinium body of 0.2 mmol/kg is injected to look for the pattern of myocardial enhancement. Late gadolinium enhancement technique (LGE) acquire images 10 to 20 minutes after gadolinium injection. These images to be taken during breath-hold using a segmented 2D or 3D inversion recovery gradient-echo, with inversion time optimized to null myocardial signal intensity.
The following three different enhancement patterns can be recognized using LGE: subendocardial, transmural, or subepicardial.11 LGE located within the perfusion territory of an epicardial coronary artery can be classified as ischemic type, and abnormal enhancement not confined to a known vascular distribution is consistent with nonischemic causes15 (Table 2).
|
MR technique |
DCM nonischemic |
Ischemic cardiomyopathy |
|---|---|---|
|
Abbreviations: DCM, dilated cardiomyopathy; LGE, late gadolinium enhancement; MRI, magnetic resonance imaging; RWMA, regional wall motion abnormalities. |
||
|
Delayed-perfusion images—LGE |
1. LGE—does not correspond to the particular coronary territory 2. Mid wall enhancement |
1. LGE—does correspond to the particular coronary territory 2. Subendocardial or transmural enhancement11 |
|
First-pass perfusion |
Normal or early increased enhancement |
In acute phase—focal perfusion defect16 |
|
Cine MRI images |
No RWMA of particular coronary territory |
RWMA |
|
Plain MRI |
Marked dilated and uniformly thinned out ventricle |
The differential thickness of the dilated ventricle. |
Ischemic Cardiomyopathy
The primary role of cardiac MRI is to differentiate ischemic versus nonischemic dilated cardiomyopathy in the evaluation of various cardiomyopathies. Cine MRI images show regional wall motion abnormality of the affected myocardium. Normal myocardium does not show enhancement, whereas the infarcted area appears bright on late-enhancement images (Fig. 1). It can be focal to arterial distribution, and subendocardium is always involved.17
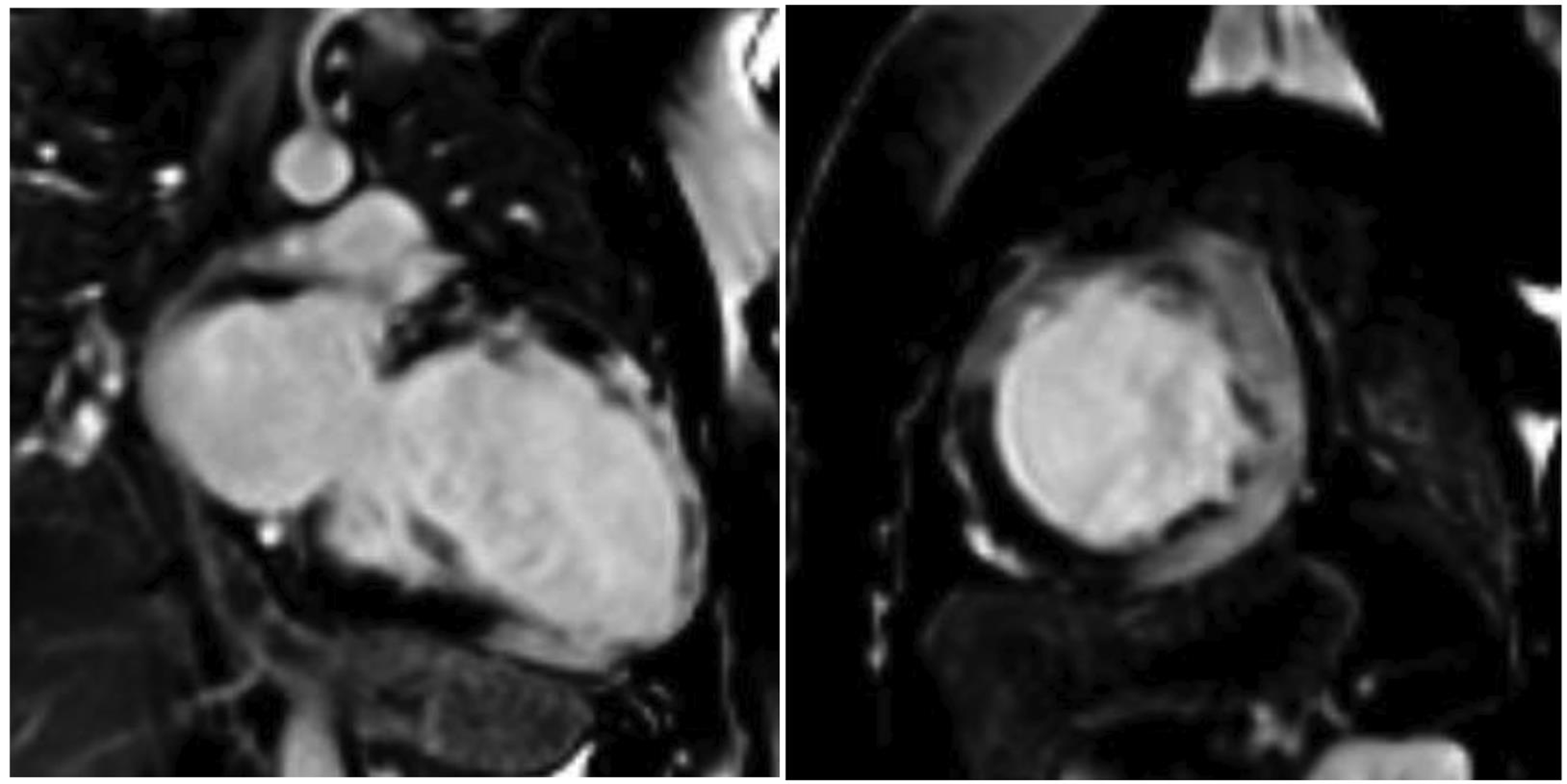
-
Fig. 1 45 Y/F with shortness of breath. Two-chamber and short-axis views are showing a dilated left ventricle, with transmural late gadolinium enhancement in anterior, lateral, and inferior walls, suggestive of infarction. Ejection fraction of 16%.
Fig. 1 45 Y/F with shortness of breath. Two-chamber and short-axis views are showing a dilated left ventricle, with transmural late gadolinium enhancement in anterior, lateral, and inferior walls, suggestive of infarction. Ejection fraction of 16%.
Both acute and chronic infarctions can enhance. In acute infarction, as there is myocardial cell membrane disruption, contrast enters into the cell. In chronic infarction, the contrast remains in the interstitium.18 Reimer and Jennings identified a pattern in late gadolinium enhancement of myocardium, that is, subendocardial extending to epicardium and transmural involvement resulting in wall motion abnormality.
Nonischemic Dilated Cardiomyopathy
Patients with idiopathic DCM either show no enhancement (Fig. 2) or linear mild myocardial enhancement (Fig. 3).However, McCrohon et al showed different patterns of enhancement. At 28%, it was a linear mild myocardial pattern, subendocardial pattern at 13%, and no enhancement at 59%.11 In asymptomatic patients or patients with mild symptoms, the presence of LGE was a predictor of adverse cardiac events.
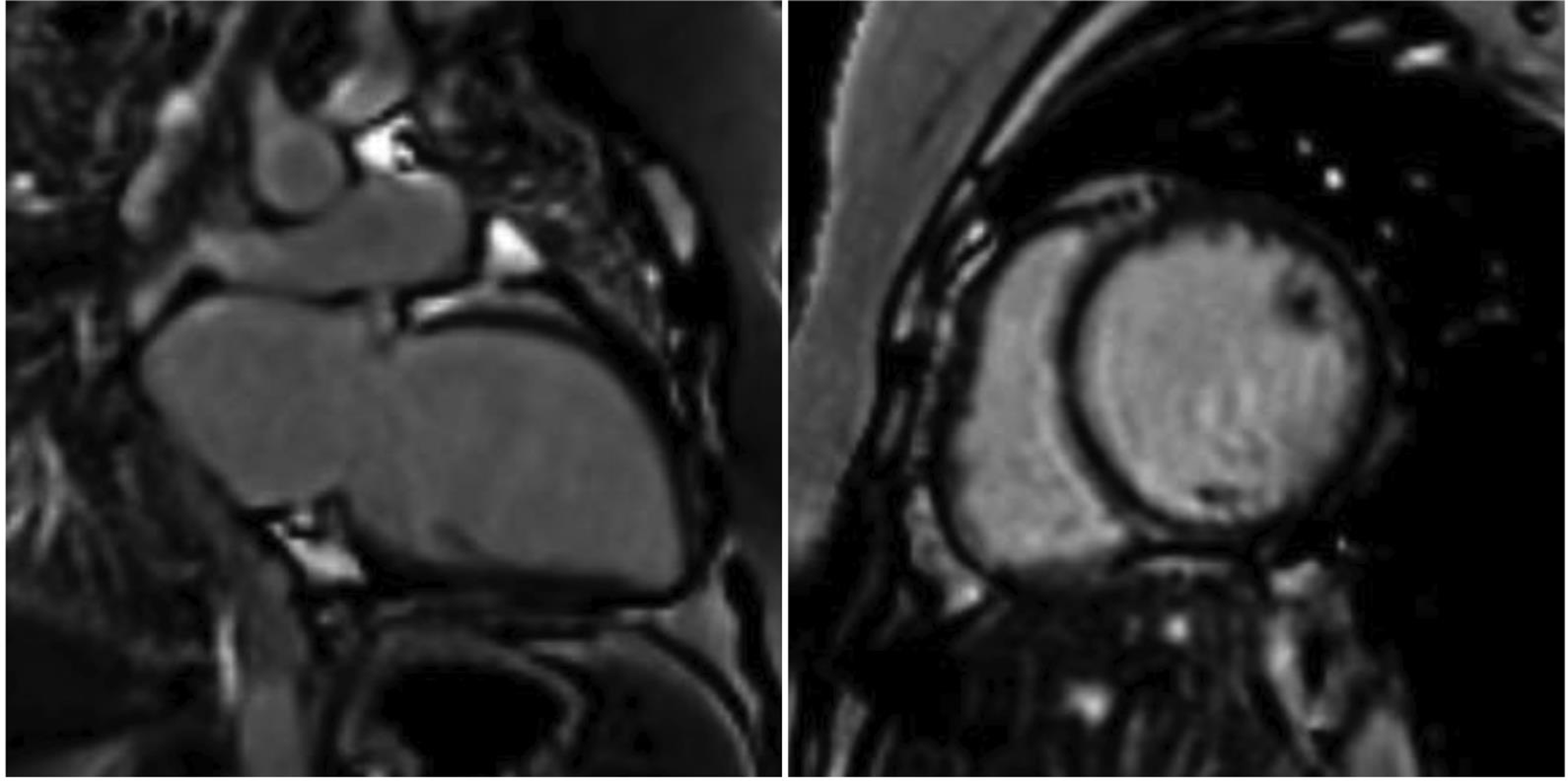
-
Fig. 2 20 Y/F with breathlessness. Two-chamber and short-axis views are showing a dilated left ventricle, with no late gadolinium enhancement in the myocardium to suggest fibrosis. The ejection fraction was 20%.
Fig. 2 20 Y/F with breathlessness. Two-chamber and short-axis views are showing a dilated left ventricle, with no late gadolinium enhancement in the myocardium to suggest fibrosis. The ejection fraction was 20%.
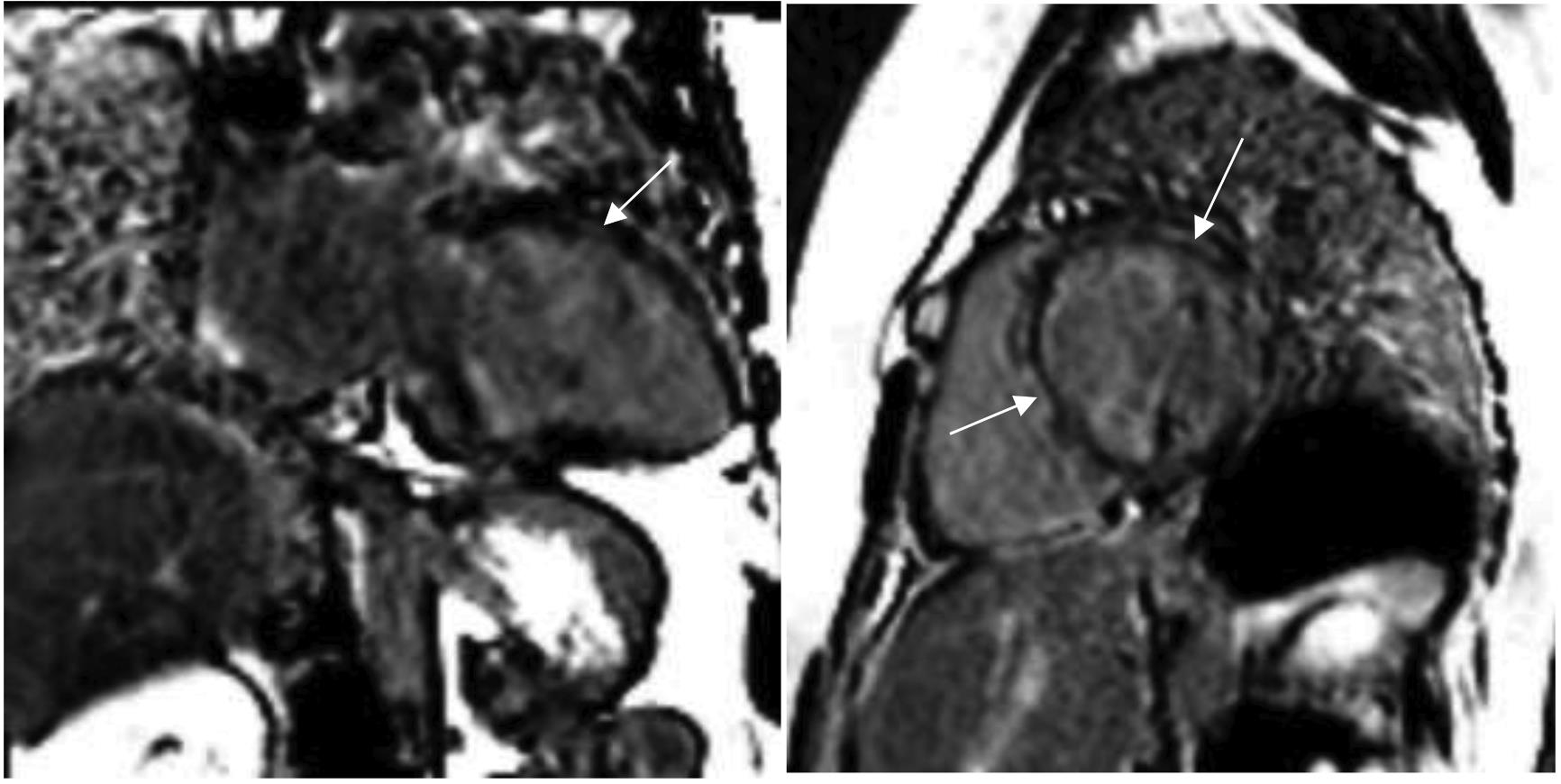
-
Fig. 3 29 Y/F with breathlessness. Two-chamber and short-axis views are showing mid myocardial (dashed arrows) and subepicardial (white arrows) late gadolinium enhancement in the anterior and apical wall, suggesting fibrosis. The ejection fraction was 26%.
Fig. 3 29 Y/F with breathlessness. Two-chamber and short-axis views are showing mid myocardial (dashed arrows) and subepicardial (white arrows) late gadolinium enhancement in the anterior and apical wall, suggesting fibrosis. The ejection fraction was 26%.
Viral Myocarditis
Coxsackie virus B3 (CBV3) is considered the dominant viral etiological agent affecting the heart muscle Fig. 3.19 Other frequently detected viral genomes are enterovirus, adenovirus, parvovirus B19 (PVB19), human herpes virus V6, and Epstein–Barr virus (EBV).20
Eighty percent of cases of myocarditis improve but 20% of cases may progress to DCM (33) due to a direct cytotoxic effect. Apart from reduced EF and regional wall motion abnormality, contrast-enhancement pattern, to some extent, helps in differentiating various viruses affecting the myocardium (Table 3). T2-weighted image depicts edema and inflammation due to myocarditis in the myocardium.21
|
Abbreviations: HHV6, human herpes virus 6; PVB19, parovirus B19. |
|
|
Area of contrast enhancement |
Type of viral myocarditis |
|
Lateral free wall |
PVB19 myocarditis |
|
Mid wall of the interventricular septum |
1. HHV6 myocarditis 2. Drug toxicity 3. Alcohol abuse11 |
In acute myocarditis, there are myocardial segments with or without LGE. T2 relaxometry distinguishes between these myocardial segments, representing an inflammatory process. So, T2 relaxometry is useful to detect the global myocardial inflammatory process during the acute phase. This is important as it is correlated with biochemical severity of myocarditis22 and planning for immunosuppressive therapy.
CE is useful to differentiate myocarditis from infarction. In addition, it gives a clue for the underlying type of virus which causes myocarditis (Fig. 3).23 The MRI findings during the acute phase of illness persist for a longer time.24 Clinical improvement in patients with myocarditis does not correlate with CMR findings and may persist for long in patients with myocarditis, as shown by Mahrholdt et al.23
3D MRI
The 3D dataset available with MRI provides a more precise and reproducible method for quantifying ventricular volumes, mass, and function in patients with DCM.
MR Spectroscopy
This method assesses cardiac metabolism noninvasively. There is no need for any contrast for this study. Hydrogen spectroscopy provides information about myocardial cellular triglyceride levels. Phosphorus spectroscopy measures myocardial energetics.25
Risk Stratification of Cardiomyopathy Using MRI
As the distinction between idiopathic DCM and ischemic cardiomyopathy is possible by MRI, which has a different prognosis, MRI is used to risk stratify the cardiomyopathy (Table 4).
|
Finding |
Implication |
|---|---|
|
Abbreviations: DCM, dilated cardiomyopathy; ICD, implantable cardioverter defibrillator; LGE, late gadolinium enhancement; MRI, magnetic resonance imaging. |
|
|
The presence of mid wall striae of tissue enhancement (may cause inducible ventricular tachycardia) |
Candidate for ICD |
|
Presence of LGE |
Predictor of sudden cardiac death |
Limitations
Like any other general contraindication to MRI study, patients with renal failure, pacemakers, etc., cannot undergo this examination. Although CMRI has several advantages over other diagnostic modalities in identifying the pathology, it experiences certain limitations in differentiating various nonischemic causes of dilated cardiomyopathy.
Conclusion
Cardiac MRI has evolved as an important diagnostic tool in the evaluation of patients with cardiomyopathy over the course of a decade. It is crucial to differentiate between ischemic and nonischemic DCMs and various conditions leading to nonischemic DCMs.
Acknowledgment
We thank our resident Sravan for helping us write this article.
Conflict of interest
The author declares that there is no conflict of interest in publishing this article.
References
- Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the definition and classification of cardiomyopathies. Circulation. 1996;93(05):841-842.
- [Google Scholar]
- Pathophysiology of Heart Disease: A Collaborative Project of Medical Students And Faculty. Baltimore MD Wolters Kluwer/Lippincott Williams & Wilkins.
- [Google Scholar]
- The importance of cardiac MRI as a diagnostic tool in viral myocarditis-induced cardiomyopathy. Neth Heart J. 2009;17(12):481-486.
- [Google Scholar]
- CMR image acquisition protocols. Society for Cardiovascular Magnetic Resonance 2008 June 3
- [CrossRef] [Google Scholar]
- A pattern-based approach to assessment of delayed enhancement in nonischemic cardiomyopathy at MR imaging. Radiographics. 2009;29(01):89-103.
- [Google Scholar]
- Cardiac amyloidosis: MR imaging findings and T1 quantification, comparison with control subjects. J Magn Reson Imaging. 2007;25(06):1283-1287.
- [Google Scholar]
- The role of echocardiography in guiding management in dilated cardiomyopathy. Eur J Echocardiogr. 2009;10(08):iii15-iii21.
- [Google Scholar]
- Differentiation of heart failure related to dilated cardiomyopathy and coronary artery disease using gadolinium-enhanced cardiovascular magnetic resonance. Circulation. 2003;108(01):54-59.
- [Google Scholar]
- Correlation between left ventricular diastolic function and ejection fraction in dilated cardiomyopathy using magnetic resonance imaging with late gadolinium enhancement. Circ J. 2009;73(10):1939-1944.
- [Google Scholar]
- Impact of cardiac magnetic resonance imaging in non-ischemic cardiomyopathies. World J Cardiol. 2016;8(02):132-145.
- [Google Scholar]
- Delayed enhancement cardiovascular magnetic resonance assessment of non-ischaemic cardiomyopathies. Eur Heart J. 2005;26(15):1461-1474.
- [Google Scholar]
- Cardiac sarcoidosis: spectrum of MRI features. AJR Am J Roentgenol. 2005;184(01):249-254.
- [Google Scholar]
- MRI in the assessment of non ischemic myocardial diseases. Eur J Radiol. 2012;81(07):1546-1548.
- [Google Scholar]
- Cardiomyocyte hypertrophy, oncosis, and autophagic vacuolization predict mortality in idiopathic dilated cardiomyopathy with advanced heart failure. J Am Coll Cardiol. 2011;57(14):1523-1531.
- [Google Scholar]
- Molecular biology and pathogenesis of viral myocarditis. Annu Rev Pathol. 2008;3:127-155.
- [Google Scholar]
- A prospective study of biopsy-proven myocarditis: prognostic relevance of clinical and aetiopathogenetic features at diagnosis. Eur Heart J. 2007;28(11):1326-1333.
- [Google Scholar]
- Diagnostic performance of cardiovascular magnetic resonance in patients with suspected acute myocarditis: comparison of different approaches. J Am Coll Cardiol. 2005;45(11):1815-1822.
- [Google Scholar]
- Myocardial edema in acute myocarditis: relationship of T2 relaxometry and late enhancement burden by using dual-contrast turbo spin-echo MRI. Int J Cardiovasc Imaging. 2017;33(11):1789-1794.
- [Google Scholar]
- Presentation, patterns of myocardial damage, and clinical course of viral myocarditis. Circulation. 2006;114(15):1581-1590.
- [Google Scholar]
- Viral myocarditis and dilated cardiomyopathy: mechanisms, manifestations, and management. Postgrad Med J. 2001;77(903):4-10.
- [Google Scholar]







